by A.S. Baranov, O.F. Chernova, N.Yu. Feoktistova, and A.V. Surov
Presented by Academician E.I. Vorob'eva
September 24, 2009
Received October 1, 2009
DOI: 10.1134/S0012496610020134
Original Russian Text © A.S. Baranov, O.F. Chernova, N.Yu. Feoktistova, A.V. Surov, 2010, published in Doklady Akademii Nauk, 2010, Vol. 431, No. 4, pp. 559–562.
ISSN 0012 4966, Doklady Biological Sciences, 2010, Vol. 431, pp. 117–120. © Pleiades Publishing, Ltd., 2010.
NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHT
YOU ARE REQUIRED TO READ THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
The term ectopia (heterotopia, tissue aberration) means the appearance of morphological structures in regions of the body where they are untypical; e.g., three cases of slight hair growth in the human oral cavity (in persons that had no hypertrichosis) have been described [1–3]. Histological analysis has shown the slightly keratinized gingival epithelium contains deep hollows, through which “hairlike structures” are likely to grow into the oral cavity, and follicles that somewhat differ from those of normal skin hair. Medical examination of these three patients did not show any disease with which oral hair could be related (all the three persons proved to be physically healthy); therefore, it was assumed that oral hair growth was caused by mutations affecting the gingival tissue [3].
It is known that some regions of the oral mucosa are more or less keratinized in different mammals. This is why gingival epithelium is capable of producing derivatives, such as teeth, sebaceous glands, and hair. Keratinization of the epithelium of the oral cavity may be induced by mechanic injury during chewing of food. In many rodent species, the buccal mucosa has cutaneous cavities sparsely covered with thin, short hairs associated with hypertrophied sebaceous glands [4–6]. However, hair had never been found on the gingival or palatal mucosa before our study.
When preparing the skulls of hamsters Phodopus campbelli from the vivarium of the Severtsov Institute of Ecology and Evolution of the Russian Academy of Sciences, we found an unusual structure, namely an extremely well developed of gingival and parodontal pouches (hereinafter, pouches), cleftlike spaces between the buccal surface of the tooth crown and the gingival margin filled with “brushes” of vertical kerati nous outgrowths, which later proved to be true hairs. Similar structures were found in some other rodents kept in the vivarium, namely Ph. sungorus and Mus musculus (table). At the same time, they were absent in Ph. robrovskii, a closely related species of the same genus, as well as in Mesocricetus auratus and Rattus norvegicus. All examined rodents received the standard diet developed in the Severtsov Institute of Ecology and Evolution; the food contained oats and compound food for rodents.
The gingival and parodontal pouches were more or less developed in all examined Ph. campbelli and Ph. sungorus. They were usually located at the internal or buccal side of the maxillary tooth row, but they were sometimes also found at the mandibular teeth and on the palate behind or in front of the tooth row. The number and sizes of these pouches considerably varied in different species, in animals of different sexes and ages, and in different parts of the palate in the same individual. Some of them were as deep as the tooth roots; in this case, the tooth row was shifted and the teeth became loose. Some of these pouches contained single hairs; others, thick bundles of colorless or pigmented hairs reaching as high as the chewing surface of the teeth. Sometimes, the tooth row was surrounded with a regular brush of hair bundles on both sides. The hairs grew vertically and had sharp ends, often covered with lumps of a mucous–sebaceous substance. There were no symptoms of gingivitis or other inflammations on the gums or palate. At the same time, we found that the bone tissue at these places was sometimes destroyed and richly perforated.

The occurrence of animals with gingival pouches (percent of the total number of examined animals)
In different individual Ph. campbelli, pouches containing hair were more often found on the maxilla and less often, on the mandible. On the maxilla, they were located on the right and on the left of the tooth row, usually in the segment between M1 and M3. They number varied, in different individuals, from one to five; the thickness of the hair bundles varied from several hairs to a dense brush tightly adjacent to the tooth. Hair growth was especially pronounced in the pouches located on the internal side of the tooth row. In some cases, the pouches were located between teeth. The interdental gingival space varied from 2–3 to 0.4– 1.2 mm in width, which was inversely related to the degree of development of the pouches: if the hair brush was thick and the pouch was deep and wide, the tooth row was shifted and deformed. After removal of the soft tissues of the palate, we found that the hair bundles were mainly adjacent to the processus alveolaris of the maxillary bone. If there was a hair brush, the hairs grew from hollows in the bone lined with mucosa, and the bone was considerably perforated (Fig. 1). The number of perforations varied from three or four to eight or more; their configurations and sizes, from round (0.08–0.3 mm) to oval (0.2 × 1.5 mm). The perforated bone area varied from 0.5 × 2.5 to 2.5 × 4.0 mm, when it filled the entire space along the tooth row and a thick bone ridge (crest) ran along the central line of the maxillary bone (Fig. 2).
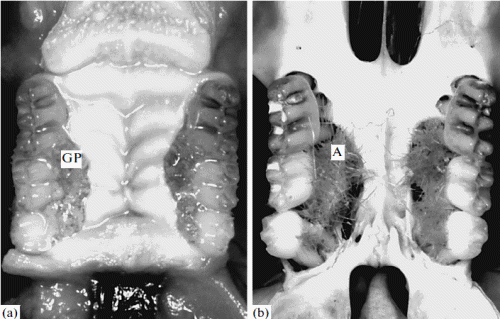
Fig. 1. (a) The external appearance of the oral cavity. Gingival pouches (GP) with thick bundles of hair growing from their mucous lining are clearly seen. (b) Perforated bone tissue of the teeth of an adult Ph. campbelli. Numerous hollows are seen. A, hair.

Fig. 2. Different parts of hairs of the gingival pouches of an adult male Ph. campbelli. The hairs are of two types: a large guard hair with a continuous pigmented medulla that has a degenerative structure and a small guard hair with a pigmented one layered ladder like medulla; the medulla reached (a) almost the very end of the hair and (b) the hair root.
The hairs filling the pouches were not foreign fragments and had not been stuffed there during grooming; they were typical hairs with well developed roots; i.e., they grew directly from the keratinized mucosal lining of the pouches. The number of hairs and the sizes of the pouches considerably varied in different individuals and in different pouches of the same individual. The hair length was usually 1–2 mm; some times, it reached 2.5 mm. Their color was mostly yellowish, but black hairs were also sometimes observed. The shape of the hair shaft was similar to the shape that we earlier described in the study on specialized hairs on the feet and midventral glands of Phodopus [6]. The shaft was of regular cylindrical shape, with a narrower stem above the root and gradual widening in the upper third. It had a sharp end, which was sometimes slightly bent. The medulla stretched along the entire shaft, from the root to the sharp end (Fig. 3a). All hairs could be divided into two types: (1) very thin (23.5 × 0.5 μm), long hairs with a poorly developed and strongly pigmented one layered ladder like medulla occupying as much as 20% of the shaft width and (2) thickened hairs (55.8 × 0.5 μm) with an unpigmented, well developed (62%) medulla, which looked degenerative: it consisted of a continuous chord or consisted of separate, irregularly oriented wide septa, with poorly developed air cavities. The pattern of the cuticle of palatal hairs was similar to that of the skin hair cover of Phodopus [6]. The cuticle was semicircular or circular, slightly flattened in the middle and at the top of the shaft compared to its base (Fig. 3b). The cuticular scales were as long as 10 μm at the base of the shaft and shorter (up to 5 μm) above the middle of the shaft. The free edges of the scales were slightly sinuate (Fig. 3d). The outer surface of some hairs lacked the cuticle, and its relief was formed by thickened edges of cortical cells oriented along the shaft (Fig. 3c), which suggested that a strong mechanical effect had destroyed their cuticular layer.
It remains unclear why these hair structures appear in the oral cavity of mammals. We may only speculate on the origin of this phenomenon. The gingival pouches may result from paradontitis and paradontosis caused by feeding on compound food in the vivarium, i.e., by a suboptimal diet. This pathology may be exacerbated by elements of the food that are absent in natural food, such as genetically modified (GM) ingredients (GM soybean or maize meal) or contaminants (pesticides, mycotoxins, heavy metals, etc.). Probably, hair growth in the gingival pouches is a protective reaction of the body suppressing the progress of gingival pathology, because the hair bundles are so dense that they prevent food from getting into the pouches and the resultant inevitable inflammation. Hair grows in the parts of the mucosa that, being affected by mechanical factors, acquire the capacity for keratinization. However, this assumption does not explain why oral hair has not been found in rats or other animals from the vivarium, including hamsters (Ph. robrovskii and M. auratus). It is unknown whether this phenomenon occurs in natural populations of rodents and, if so, how common it is.
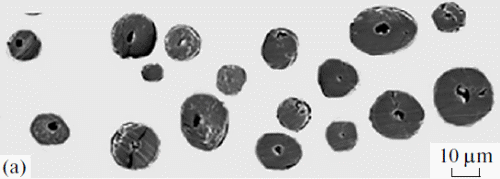
Fig. 3. (a) Cross sections and (b–d) the cuticle of hairs of the gingival pouches of an adult male Ph. campbelli.
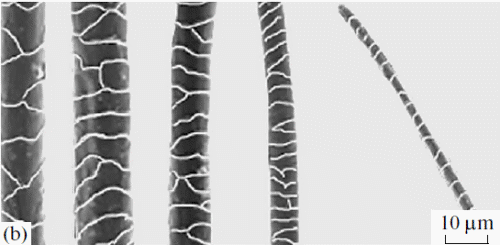
(b) A cylindrical or slightly deformed hair shaft with a central medulla canal running along the middle of the shaft or slightly shifted; the medulla is poorly developed.

(c) A destroyed cuticular layer; rough edges of cortical cells are seen.
It is noteworthy that this phenomenon has been found in representatives of phylogenetically remote taxa (rodents and humans). Further research is required to determine the cause and mechanism of the phenomenon of oral hair in mammals.
ACKNOWLEDGMENTS
We are grateful to D.M. Shchepotkin (Kol’tsov Institute of Developmental Biology of the Russian Academy of Sciences) for the assistance in preparing the photographs.
This study was supported by the Grant of the President of the Russian Federation for Leading Scientific Schools (project no. NSh 7522.2010.4), Russian Fou dation for Basic Research (project no. 09 04 000 701), and the Program of the Presidium of the Russian Academy of Sciences “Biological Diversity” (project no. NOTs 02.740.11.02.82).
REFERENCES
1. Miles, A.E.W., Proc. Roy. Soc. Med., 1960, vol. 53, pp. 527–528.
2. Baughman, R.A., Paul, D., and Heidrieh, Jr., Oral Surg. Med. Oral Pathol., 1980, vol. 49, pp. 530–531.
3. Agha Hosseini, F., Etesam, F., and Rohani, B., Med. Oral. Pathol. Oral Cir. Bucal., 2007, vol. 12, no. 5.
4. Quay, W.B., J. Mammal., 1965, vol. 46, no. 1, pp. 23– 27.
5. Sokolov, V.E. and Chernova, O.F., Kozhnye zhelezy mle kopitayushchikh (Skin Glands of Mammals), Moscow: KMK, 2001.
6. Feoktistova, N.Yu., Khomyachki roda Phodopus. Siste matika, filogeografiya, ekologiya, fiziologiya, povedenie, khimicheskaya kommunikatsiya (Hamsters of the Genus Phodopus: Phylogeography, Ecology, Physiol ogy, Behavior, and Chemical Communication), Mos cow: KMK, 2008.
