Crisis in the Hot Zone: Lessons from an outbreak of Ebola
by Richard Preston
October 19, 1992
The New Yorker, October 26, 1992 Issue
NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHT
YOU ARE REQUIRED TO READ THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
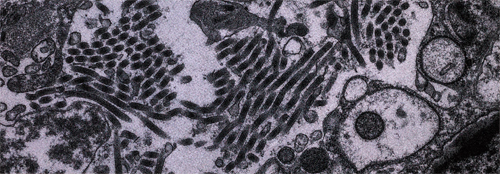
In 1989, Washington, D.C., saw an outbreak of the Ebola Reston virus, a close relative of a species that killed nine out of ten infected people in Africa, in 1976.Photograph Courtesy Thomas Geisbert / USAMRIID
The main building of the United States Army Medical Research Institute for Infectious Diseases is an essentially windowless concrete block that covers several acres at Fort Detrick, an Army base in Frederick, Maryland, fifteen miles east of Antietam. Military people call the structure the Institute, or they call it by its acronym, usamriid, drawling it as You Sam Rid. Or they call the place riid, as in getting rid of something. Vent stacks on its roof discharge filtered exhaust air from sealed biological laboratories inside the building. Fort Detrick, the envelope of usamriid, sits in rolling country on the eastern slope of the Appalachian Mountains, in the drainage of the Potomac River. The Potomac bends through oak-blanketed mountains at Harpers Ferry and enters farmland, and eventually passes near Reston, Virginia, a town outside the Washington Beltway where farms give way to business parks, and where in the eighties office buildings accreted like crystals.
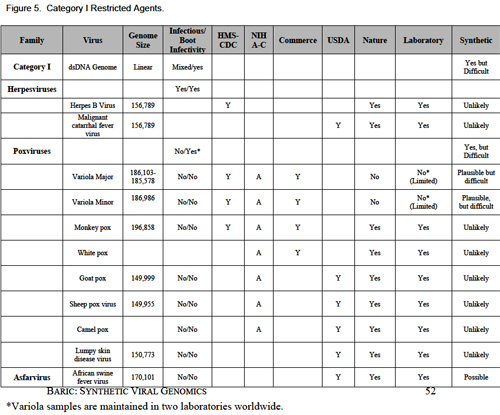
The mission of usamriid is medical defense. The Institute conducts research into ways to protect soldiers against biological weapons and natural infectious diseases. It specializes in vaccines, drug therapy, and biocontainment. That is, the Institute knows methods for stopping a monster virus before it ignites an explosive chain of lethal transmission in the human race. The laboratory suites at usamriid are maintained at four levels of biological security. The levels go from Biosafety Level 1, which is the lowest, up to Biosafety Level 4, the highest. The Biosafety Level 4 rooms contain BL-4 agents, also known as hot agents. A BL-4 hot agent is a lethal virus for which, in most cases, there is no vaccine and no cure. It is in the nature of hot agents to travel through the air: they can become airborne. The hot agents live in the hot suites in blood serum and bits of meat, frozen at -70° Centigrade. All the biocontainment laboratories at usamriid are kept under negative air pressure, so that if a leak develops air will flow into the hot rooms and out of the normal world, rather than the other way around. The Army does not publish a list of the viruses it keeps in the hot suites at usamriid, but here is a list of BL-4 viruses: Junin. Lassa. Machupo. Tick-borne encephalitis virus complex. Guanarito. Crimean-Congo. Marburg. Ebola Sudan. Ebola Zaire. Ebola Reston. If you want to shake hands with one of these viruses, you had better wear a space suit. That’s a federal rule. It holds equally at usamriid and at the Centers for Disease Control, in Atlanta, which are the only two laboratories in the United States that can handle BL-4 viruses.
To go inside a Biosafety Level 4 hot suite that contains life, first you have to strip naked. You put on surgical scrubs and then a space suit. You pull the helmet down over your head and close the suit. Then you enter an antechamber, a kind of air lock. It leads to Biosafety Level 4. Military people consider this air lock a gray zone, a place where two worlds meet. The air-lock doors are blazed with the international symbol for biohazard, a red trefoil that reminds me of a flower. I think it looks not unlike a red trillium, or toadshade. At usamriid, toadshades bloom in the gray zones.
Lieutenant Colonel Nancy Jaax is the chief of the pathology division of usamriid. She is a slender and rather beautiful woman, a doctor of veterinary medicine, forty-two years old, with curly auburn hair and green eyes. She has a brisk manner. On the job, Nancy Jaax wears a uniform consisting of green slacks and a green shirt with shoulder bars displaying the silver oak leaves of her rank. Or she wears a space suit. She is married to Colonel Gerald Jaax, who is the chief of the veterinary-medicine division at usamriid. The Army assigned Nancy Jaax and her husband to usamriid in 1979. She had just been awarded the rank of major, and she entered the pathology training program at usamriid as a veterinary-pathology resident. Pathologists at usamriid, who cut up hot tissue, are given vaccinations for lethal agents. Nancy Jaax said to me, “My vaccinations were for yellow fever, Q fever, Rift Valley—there were so many. The V.E.E., E.E.E., and W.E.E. complex, anthrax, and botulism. And, of course, rabies, since I’m a veterinarian.” She had an underlying medical condition that caused her immune system to react badly to the shots: the shots made her sick. The Army therefore stopped her vaccinations and assigned her to work in a space suit in the Biosafety Level 4 suites. “There aren’t any immunizations for most BL-4 agents, and that’s why you work in a space suit,” she explained.
In 1980, Nancy Jaax joined a group of military scientists who were performing experiments with Ebola virus on monkeys. They were infecting monkeys with Ebola and then treating them with interferon and other substances to see if the treatments stopped or weakened the disease. The purpose of the experiments was to find some chemical therapy for military personnel who might become infected with Ebola.
Ebola is one of a class of viruses known as the filoviruses. That means thread viruses. They look like spaghetti. As of this writing, the class comprises three subtypes of Ebola and a virus known as Marburg. Ebola virus is named for the Ebola River, a tributary of the Zaire (Congo) River which runs through northern Zaire. The first known emergence of Ebola Zaire—the hottest subtype of Ebola virus—happened in September, 1976, when the virus erupted simultaneously in fifty-five villages near the Ebola River. Ebola Zaire is a slate-wiper in humans. It killed eighty-eight per cent of the people it infected. Apart from rabies and the human immunodeficiency virus, H.I.V., which causes aids, this was the highest rate of mortality that has been recorded for a human virus. Ebola was spread mainly among family members, through contact with bodily fluids and blood. Many of the people in Africa who came down with Ebola had handled Ebola-infected cadavers. It seems that one of Ebola’s paths wends to the living from the dead.
Ebola victims died about a week after the onset of the first symptom, which was a headache. The Ebola patient soon breaks into a relentless fever, and then come the complications. Ebola triggers a paradoxical combination of blood clots and hemorrhages. The patient’s bloodstream throws clots, and the clots lodge everywhere, especially in the spleen, liver, and brain. This is called D.I.C., or disseminated intravascular coagulation. D.I.C. is a kind of stroke through the whole body. No one knows how Ebola triggers blood-clotting. As the strokelike condition progresses and capillaries in the internal organs become jammed with clots, the hemorrhaging begins: blood leaks out of the capillaries into the surrounding tissues. This blood refuses to coagulate. It is grossly hemolyzed, which means that its cells are broken. You are stuffed with clots, and yet you bleed like a hemophiliac who has been in a fistfight. Your skin develops bruises and goes pulpy, and tears easily, and becomes speckled with purple hemorrhages called petechiae, and erupts in a maculopapular rash that has been likened to tapioca pudding. Your intestines may fill up completely with blood. Your eyeballs may also fill with blood. Your eyelids bleed. You vomit a black fluid. You may suffer a hemispherical stroke, which paralyzes one whole side of the body and is invariably fatal in a case of Ebola. In the pre-agonal stage of the disease (the endgame), the patient leaks blood containing huge quantities of virus from the nose, mouth, anus, and eyes, and from rips in the skin. In the agonal stage, death comes from hemorrhage and shock.
People seem unable to develop protective antibodies to Ebola. You can’t fight off an Ebola infection the way you fight off a cold. Ebola seems to crush the immune system. The virus perhaps makes immunosuppressant proteins. No one knows the nature of such proteins, since there aren’t many virologists who care to study a virus for which there is no vaccine and no cure. (They don’t want the virus to do research on them.) Immunosuppressive proteins—if, indeed, they exist—would act as molecular bombs that ruin parts of the immune system, enabling the virus to multiply without opposition.
Like all viruses, Ebola and its cousin Marburg are parasites. They can copy themselves only inside a cell. Viruses need to use a cell’s equipment to reproduce. Ebola and Marburg grow promiscuously in human tissue, sprouting from cells like hair, forming tangled masses and braids and “g”s and “y”s and pigtails. Marburg-virus particles often roll up into tiny Cheerios. All filoviruses form semi-crystalline blocks inside cells, which are known as inclusion bodies. Some scientists call them bricks. The bricks may pack a cell until there’s almost nothing left of the cell but bricks: the cell bloats into a sack of bricks. Then the bricks break apart into threads of virus, and the threads push through the cell wall like grass rising from seeded loam.
A classic sign of infection by Ebola or Marburg is a certain expression that invariably creeps over the patient’s face as the infection progresses. The face becomes fixed and “expressionless,” “masklike,” “ghostlike” (in the words of doctors who have seen it), with wide, deadened, “sunken” eyes. The patient looks and sometimes behaves like a zombie. This happens because Ebola damages the brain in some way that isn’t known. The classic masklike facial expression appears in all primates infected with Ebola, both monkeys and human beings. They act as if they were already embalmed, even though they are not yet dead. The personality may change: the human patient becomes sullen, hostile, agitated, or develops acute psychosis. Some have been known to escape from the hospital.
Disseminated clotting cuts off the blood supply to tissues, causing focal necrosis—dead spots in the liver, spleen, brain, kidneys, and lungs. In severe cases, Ebola kills so much tissue that after death the cadaver rapidly deteriorates. In monkeys, and perhaps in people, a sort of melting occurs, and the corpse’s connective tissue, skin, and organs, already peppered with dead areas and heated with fever, begin to liquefy, and the slimes and uncoagulated blood that run from the cadaver are saturated with Ebola-virus particles. That may be one of Ebola’s strategies for success.
Lieutenant Colonel Nancy Jaax’s job during the Army’s 1980 experiments with Ebola was to dissect and examine monkeys that had died of the virus. Her space suit had triple pairs of gloves. First, there was an inner latex surgical glove. Over that, the suit had attached to it a heavy rubber glove. Over the rubber glove she wore another latex surgical glove. Her space suit and gloves were often splashed with blood as she cut into dead monkeys, and she regularly dipped her gloves in a pan of Envirochem—a liquid disinfectant that the Army believes is effective on viruses—to rinse away the blood. They use a buddy system in BL-4. You don’t work alone in a hot area. The buddies are trained to glance at each other’s gloves for leaks. (“The weak link is your glove,” Jaax told me. “You are handling needles, knives, and sharp pieces of bone.”) One day, Jaax’s buddy noticed a hole in Jaax’s right outer latex glove. The glove was covered with Ebola-laden blood. Jaax rinsed the glove in Envirochem and took it off, and found monkey blood inside it: the blood had run through the hole and drenched the heavy rubber glove.
Then she felt something clammy inside the heavy glove. She wondered if it was a leaker.
She rinsed her bloody glove and went into the air lock. There, still wearing her space suit, she pulled a chain to start the decontamination, or “decon,” cycle. The decon cycle took five minutes. First, a hot-water shower came on, and then came a mist of Envirochem, which washed away any blood from the exterior of her suit, while sterilizing it. She stepped into a tub of Envirochem, bent over, put her hands in the tub, and scrubbed her booties and gloves with a brush. (In the old days, the Army’s air-lock showers ran with Lysol. It kills germs, as advertised, but it made some people itch.) Then a final water shower came on and stopped. Nancy Jaax left the air lock and entered a staging area, where she stepped out of her space suit, withdrawing her latex-gloved hands from the suit’s heavy gloves. As her right hand came out of the suit, she saw it was red—bloody. The suit’s heavy glove had been a leaker.
The blood had smeared the innermost latex glove, right against her skin. Her heart pounded, and her stomach turned over. “I got that oogh feeling. That feeling you sometimes get when you work with these agents,” she said. “I went, ‘Oh, shit. What now? Oh, Jesus. What do I have to do now?’ ” On her right hand, under the last glove, she had an open cut in her skin. She does all the cooking for her family; she had cut herself with a paring knife while slicing vegetables, and had covered the cut with a Band-Aid. The question was whether any blood had penetrated the last glove to the Band-Aid and the cut. If so, it would amount to a death warrant. Five or ten virus particles suspended in a microscopic droplet of blood could easily slip through a pinhole in a surgical glove, and that would probably be enough to start a fatal infection. At usamriid, there is a group of pressurized hospital rooms designed so that patients can be treated by nurses and doctors wearing space suits. The place is an isolation hospital, and they call it the Slammer. Nancy Jaax began to wonder if she would end up in the Slammer by nightfall. She and her husband have two children. She did not want to break with Ebola virus in the Slammer and never see her children again. She dipped her last, bloody glove in Envirochem, and went over to a sink, and removed the glove. She put it under a faucet and filled it with water, like a water balloon. It held. No leaks. “This incident came into the category of close call,” she said to me.
Nancy Jaax continued with the experiment, and all the monkeys that had been infected with Ebola died; the drugs had no effect on the course of the disease. She kept two control monkeys—healthy monkeys—apart from the others, in separate cages inside the hot suite. Then both control monkeys died of Ebola. They had not been injected with virus, and their cages were on the far side of the room from those of the sick monkeys. “So the question is: How did they get it?” Lieutenant Colonel Nancy Jaax said to me. “They probably got it from aerosolized droplets from the sick monkeys. That was when I knew that Ebola could spread through the air.”
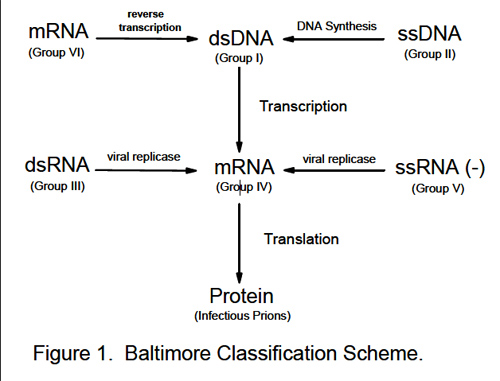
Avirus is a small capsule consisting of membranes and proteins. The capsule holds one or more strands of RNA or DNA that contain the software program for making a copy of the virus. The virus penetrates a cell wall, and the capsule breaks apart inside the cell, releasing the strands of genetic material, which take over the cell and force it to make copies of the virus. Eventually, the cell gets pigged with virus, and pops. Or viruses can bud through a cell wall like sweat coming off a drip hose. In either case, viruses tend to kill cells. If they kill enough cells, or if they kill a class of cell that the host needs for survival, then the host dies. Viruses that kill their hosts do not themselves survive. It is in the virus’s best interest to let the host live, but accidents happen. Some biologists classify viruses as “life forms”—ambiguously alive. Bacteria and cells are always humming with activity, enzymatic processes. Viruses that are outside cells merely sit there; nothing happens. But when they get inside a cell they switch on and begin to replicate. Viruses can seem alive when they multiply, but in another sense they are molecular machines—obviously non-living, strictly mechanical, no more alive than a jackhammer. Compact, logical, hard, engineered by the forces of evolution, and totally selfish, the viral machinery is dedicated to making copies of itself—which it can do on occasion with radiant speed.
Viruses are not easy to see, even with an electron microscope. Here is a way to imagine the size of a virus. Consider the island of Manhattan, shrunk to this size:
▌
This shrunken Manhattan could easily hold nine million common-cold viruses. If you made an aerial reconnaissance of it with an electron microscope, you would see little figures milling like the lunch crowd on Fifth Avenue. Viruses can be purified and concentrated into crystals. Packed in a crystalline layer, shoulder to shoulder and only one virus deep, a hundred million polio viruses could cover the period at the end of this sentence. There could be a thousand Giants Stadiums of viruses sitting on that period—two hundred and fifty Woodstocks of viruses, a third of the population of the United States, sitting on that period—but you wouldn’t know it without a scope.
In 1892, a Russian scientist named Dimitry Ivanovsky studied a disease of tobacco leaves which gives them white spots. He passed the juice of sick leaves through extremely fine filters, and when he injected healthy plants with the filtered juice they got sick and developed white spots. Ivanovsky concluded that some very small agent was causing the disease, but he didn’t know whether it was a toxic chemical or a living thing. In 1898, Martinus Beijerinck, a Dutch botanist, proved that Ivanovsky’s virus was a replicative infectious agent. It has since come to be called tobacco-mosaic virus. In 1900, the United States Army discovered the first human virus—the yellow-fever agent. That was the work of Walter Reed and his team. The Army has tracked viruses from the beginning.
There is no fossil record in rocks to indicate that viruses existed before the late nineteenth century, when tobacco-mosaic virus was first noticed. Fossils of bacteria have turned up in rocks that are more than three billion years old, but no fossils of viruses have ever been found. Nevertheless, viruses are obviously ancient, and perhaps primeval. They are molecular sharks, a motive without a mind. They have sorted themselves into tribes, and they infect everything that lives.
The human immunodeficiency virus, or H.I.V., is a not very infectious but lethal Biosafety Level 2 or 3 agent, which most likely emerged from the rain forests of Central Africa. You don’t need to wear a space suit while handling blood infected with H.I.V. During the nineteen-seventies, the virus fell like a shadow over the human population living along the east-west highway that links Kinshasa, in Zaire, with Mombasa, in Kenya. The emergence was subtle: the virus incubates for years in a human host before it kills the host.
A zoonotic virus is a virus that lives naturally in an animal and can infect human cells, perhaps mutating slightly in the course of passage, which enables the virus to start a chain of infection through human hosts. For example, H.I.V.-2 (one of the two major strains of H.I.V.) may be a mutant zoonotic virus that jumped into us from an African monkey known as the sooty mangabey, perhaps when monkey-hunters touched bloody tissue. No one really knows where H.I.V. came from. H.I.V.-1 (the other strain) may have jumped into us from chimpanzees, or it may be a human virus that has been in our species for ages,circulating in some isolated group of people in Central Africa. As outsid-ers came into the area, aids came out, and passed into the general human population.
The emergence of aids appears to be a natural consequence of the ruin of the tropical biosphere. Unknown viruses are coming out of the equatorial wildernesses of the earth and discovering the human race. It seems to be happening as a result of the destruction of tropical habitats. You might call aids the revenge of the rain forest. aids is arguably the worst environmental disaster of the twentieth century, so far. Some of the people who worry in a professional capacity about viruses have begun to wonder whether H.I.V. isn’t the only rain-forest virus that will sweep the world. The human immunodeficiency virus looks like an example rather than a culminating disaster. As lethal viruses go, H.I.V. is by no means nature’s preëminent display of power. The rain forest, being by far the earth’s largest reservoir of both plant and animal species, is also its largest reservoir of viruses, since all living things carry viruses. Just how large the tropical reservoir of viruses is no one knows, but here is one way to consider the question. The earth is estimated to contain between three million and thirty million species of plants and animals. Most of the species are fungi, insects, and non-insect arthropods, such as ticks and mites, and the bulk of them live in tropical forests. Viruses often adapt to one or two species. For example, human beings carry more than a hundred different cold viruses that are adapted almost exclusively to the human host. If we suppose that every species carries one virus exclusively adapted to it, then there may be from three to thirty million strains of viruses. Possibly the number of virus strains is much larger than that—perhaps a hundred million—but nobody has ever tried to count them.
When an ecosystem suffers degradation, many species die out and a few survivor-species have population explosions. Viruses in a damaged ecosystem can come under extreme selective pressure. Viruses are adaptable: they react to change and can mutate fast, and they can jump among species of hosts. As people enter the forest and clear it, viruses come out, carried in their survivor-hosts—rodents, insects, soft ticks—and the viruses meet Homo sapiens. Here are the names of some emerging viruses: Lassa. Rift Valley. Oropouche. Rocio. Q fever. V.E.E. Guanarito. Ross River. Monkeypox. Dengue. Chikungunya. Hantaan. Machupo. Junin. The rabies-like strains Mokola and Duvenhage. Le Dantec. Human immunodeficiency virus—which might have been called Kinshasa Highway, if it had been noticed earlier—is considered an emerger, since its penetration of the human race is incomplete and is still happening explosively, with no end in sight. The Kyasanur Forestvirus. The Semliki virus. Crimean-Congo. Sindbis. O’nyong-nynong. Marburg. Ebola. Most of them—but not all—come from tropical forests or tropical savannas. When a virus that lives in some nonhuman host is about to crash into the human species, the warning sign may be a spatter of breaks—disconnected emergences, at different times and places. I tend to think of rats leaving a ship. The presence of international airports puts every virus on earth within a day’s flying time of the United States.
Reston, Virginia, is near Washington. The town has an active chamber of commerce and a visitors’ center designed to lure high-technology businesses to the area. Along the Leesburg Pike, a commuter route that funnels traffic to Washington, you see developments of executive homes. The homes are pseudo-Victorians, with unused porches, and stick-built neo-Georgians, with false-brick fronts and a Baby Benz parked in a semicircular carriageway. You also see the occasional bungalow with cardboard stuffed in a broken window and a Harley in the driveway. The town of Reston is bisected by the Dulles Access and Toll Road, which connects Dulles airport with Washington. Not far from the Dulles Access Road in Reston is a small business park. Until recently, a company called Hazleton Research Products had a monkey house in a one-story building in the business park. It was known as the Reston Primate Quarantine Unit. Hazleton Research Products sells animals for research; it is a division of Corning Incorporated. Hazleton was importing monkeys from the tropics and bringing them through J.F.K. International Airport to the Reston Primate Quarantine Unit. Each year, about sixteen thousand wild monkeys are imported into the United States, to be used as laboratory animals. Federal regulations require that imported monkeys be held in quarantine for at least thirty-one days before they are shipped anywhere else in the United States. This is to prevent the spread of infectious diseases that could kill other primates, including laboratory workers.
Dan Dalgard, doctor of veterinary medicine, is the principal scientist at Hazleton Washington, which has its offices on the Leesburg Pike, in Vienna, next to Reston. Dan Dalgard has an international reputation as a knowledgeable and skilled veterinarian who specializes in primate husbandry, and he understands monkey behavior and monkey diseases. He is a calm, blunt man in his late fifties. He wears glasses, and he has a square, pleasant face. On evenings and weekends, he repairs antique clocks as a hobby. He likes to use his hands and his mind to figure out how a broken complicated system can be fixed. Dalgard sometimes has longings to leave veterinary medicine and immerse himself in clocks.
On Wednesday, October 4, 1989, Hazleton accepted a shipment of a hundred wild monkeys from the Philippines. The shipment originated on the island of Mindanao, at a Philippine monkey-export company. The monkeys were macaques, and the species was Macaca fascicularis. Zookeepers call it the crab-eating macaque. It is a common monkey that lives along rivers and in mangrove swamps in Southeast Asia, and it is often used as a laboratory animal. It eats fruit, crabs, insects, and small pieces of clay. A crab-eating macaque will snatch a crab out of the water and quickly rip its claws off and throw them away before devouring the rest of the crab. Sometimes a crab-eating macaque isn’t quick enough with the claws, and when the monkeys are on a feeding bout in a mangrove swamp at low tide you can occasionally hear shrieks when a crab fastens on a monkey. The crab-eating macaque has brown eyes, pointed ears, tawny fur, and a long tail. As monkeys go, crab-eating macaques have a calm temperament, provided that you don’t stare at them. Any monkey thinks staring is rude, and the crab eater will respond on the same level, screaming “Kra, kra! ” and hurling its feces at you.
The Philippine monkeys arrived at J.F.K. and were taken by truck to Hazleton’s Reston Primate Quarantine Unit. The monkeys were kept in stainless-steel cages in windowless rooms, under artificial lights, and were fed monkey biscuits. The Reston quarantine rooms were designated by letters ofthe alphabet, from “A” through “L.” The Philippine monkeys were put in Room F. The ventilation system recirculated some air in common through the rooms, so that the monkeys were breathing one another’s air.
By the first of November, twenty-seven monkeys had died. That was more than usual for a shipment of wild monkeys. Dan Dalgard performed necropsies on the ones that had died, and concluded that they were being killed by dysentery and pneumonia. These diseases are not uncommon in wild monkeys. A week later, on Monday, November 6th, another shipment of crab-eating macaques arrived, making a total of about five hundred monkeys in the quarantine unit, all crab-eating macaques from the Philippines. But by November 10th Dalgard had begun to suspect that some of his monkeys might be dying of simian hemorrhagic fever, or S.H.F., a virus that is lethal to monkeys but does not cause clinical disease in humans. (It infects people but doesn’t make them sick.) The possibility worried Dalgard, because S.H.F. is highly contagious in monkeys, and can wipe out a colony.
He began sacrificing monkeys that appeared sick, by injecting them with overdoses of an anesthetic, and then he opened them up. He found that their spleens were enlarged—a classic sign of simian hemorrhagic fever. But monkeys infected with S.H.F. typically die sneezing blood or with other evidence of hemorrhaging, and Dalgard hadn’t seen any of these signs in the monkeys that died before November 10th. The monkeys had simply stopped eating and died of shock. The focus of the infection was Room F, where most of the monkeys had perished. The disease gave Dalgard an eerie feeling, and prompted him to keep a diary. Of the monkeys that had died in Room F he wrote:
Many of the animals were in prime condition and had more abdominal and subcutaneous fat than is customary for animals arriving from the wild. The diagnosis at this time was continuing to point more strongly toward S.H.F. but the slow progression [of the disease] and the lack of the hemorrhagic component confused the diagnosis.
He decided to take the mystery to the United States Army Medical Research Institute for Infectious Diseases, where he knew about a virologist named Peter Jahrling, who had done work on S.H.F. He described to Jahrling the illness that was burning through his monkeys, and he sent some blood and tissue samples to Jahrling. Some of the samples came from a monkey known as O53, which had lived in Room F. Jahrling froze some of the tissues and placed them in a Biosafety Level 3 containment room. This level is kept under negative pressure, but you don’t need to wear a space suit inside it.
One way to identify a virus is to make it multiply inside living cells in a flask. You drop a very small sample of the virus into the cells, and as the virus spreads through the cells extraordinary numbers of virus particles are produced. You can then look at them under a microscope, or you can put different kinds of fluorescent antibodies—immunity proteins—in the virus culture. These antibodies attach themselves to infected cells and glow under ultraviolet light, and the antibody that makes cells glow tells you which particular virus you have in the flask.
A civilian technician named Joan Rhoderick cultured the unknown monkey virus from the liver of Monkey O53. She ground up a bit of the liver with a mortar and pestle, and dropped some of the resultant mush into flasks that contained a living strain of cells from the kidney of a green monkey. Joan Rhoderick wore a surgical mask and rubber gloves but not a space suit, and she worked with the samples kept in a safety cabinet that pulls air away from the samples and through a filter.
John Rhoderick and Peter Jahrling looked at slices of liver and spleen from Monkey O53, and Jahrling gave a presumptive diagnosis of simian hemorrhagic fever to Dan Dalgard. At this point, Dalgard felt that he had no choice but to sacrifice all the monkeys in Room F in order to halt the spreading disease. If those monkeys were infected with S.H.F., they would die anyway, and if they weren’t sacrificed the disease could spread to other rooms, killing more monkeys. Dalgard and an assistant, wearing surgical masks and rubber gloves, euthanized all the monkeys in Room F on November 16th—some seventy monkeys in all. They gave the monkeys injections of an anesthetic. Dalgard opened ten of the corpses to see what he could see, and sent everything to an incinerator.