Chapter 22: The Special Virus Cancer Program
THE next morning, figuring I had learned as much about Marburg as the experts (not including Preston) chose to reveal, I decided to look for the few Congressional Record citations I couldn't find in other libraries. This time, with the Davis Library "basement pass" in hand, I approached a tall weighty fellow standing behind the government document librarian's desk.
"Hi. I'm looking for these Congressional Record references," I said, pointing to a few computer printouts.
"The Congressional Records are in the basement," he replied in a mildly inconvenienced tone. Then with a stuffy air of library science about him, he added, "I'll have to go down to get you a few at a time."
"I have my own pass," I proudly displayed. "How 'bout if you just show me where to look?"
"Fine. Follow me," he conceded.
I negotiated the department's swinging gate, passed through a set of open doors, and skipped down a flight of stairs in hot pursuit of my guide. As I entered the huge documents room, the librarian, who had gained about ten yards on me, pointed ahead and said, "The Congressional Records are over there."
Then, something very strange happened. Customarily, at least out of courtesy, I would have followed the man. But suddenly I felt myself entering what I can only describe as a "twilight zone." It was as if time and everything else stood still. My intuition told me to turn right. Without hesitating or missing a step I simply obeyed leaving my guide behind.
I had heard of uncommon intuitive leaps that led scientists to make important discoveries. I often used the example of Einstein who explained his theory of relativity (E=MC2)as a breakthrough that came to him during twilight sleep. I suppose, in retrospect, this experience was like that for me -- somewhat of an intuitive breakthrough -- except I wasn't sleeping.
Instinctively, I was being drawn in another direction; like metal to a distant magnet. I simply went with the force. Concerned my behavior, which must have seemed rude and impulsive, would further inconvenience the librarian, I blurted, "You don't mind if I have a look over here?"
That, along with my abrupt departure, obviously startled him. Stumbling for words, he replied, "You're not going to find anything over there." Then when he realized my course was set, he half obligingly advised, "That's not how you conduct research. You're likely to miss what it is you're looking for."
"I appreciate that," I said, but clearly paid no heed. I just continued along my merry way down the isle to the source of my beguilement.
The librarian, forced to concede, said, "I'll get your Congressional Records." We parted, him leaving me to prove myself wrong.
I wandered auspiciously for about fifteen yards. A voice inside said, "Turn left." I obeyed and then began to slow. My neck suddenly cranked to the left. Instantly and intently my eyes began scanning the unmarked shelves for something I knew would be there. I just wasn't sure what it was. Momentously, my focus turned knee high, and roamed title by title for a treasure. Then, suddenly, there it was. A faded red-covered book with white letters bleeding through. The words Special Virus Cancer Program touched my soul.
I grabbed the title in total disbelief. Completely amazed at what had just happened, all I could say was, "Wow! This is just what I'm looking for!"
Seconds later, the librarian walked up carrying three volumes of Congressional Record's. "Here's what you wanted," he said as he laid the texts down on a desk. ''I'm glad you found what you wanted." Then he turned and walked away.
Still dazed as he disappeared from sight, I returned, 'Thanks a lot, I really appreciate your help."
For months, I had read dozens of authors acknowledging the NIH's "Special Virus Cancer Program." Now, here was the book on it (see fig. 22.1).1 The text, covered the period from 1970 to 1971 -- the critical period during which I knew Gallo and company had created numerous AIDS-like retroviruses. I thumbed through the 383-page NCI "Progress Report #8" and realized it detailed virtually all the important investigations conducted in support of Nixon's "war on cancer." It identified all the contracting agencies; summarized their grant proposals; and provided the names, addresses, and even telephone numbers of the project directors, officers, and study consultants for the entire operation.
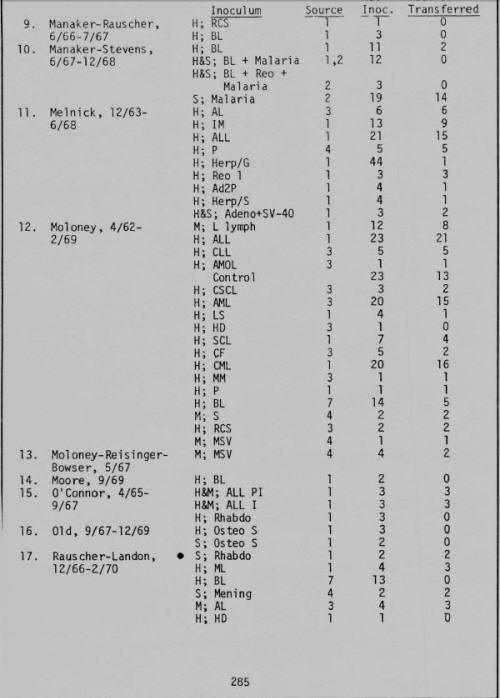
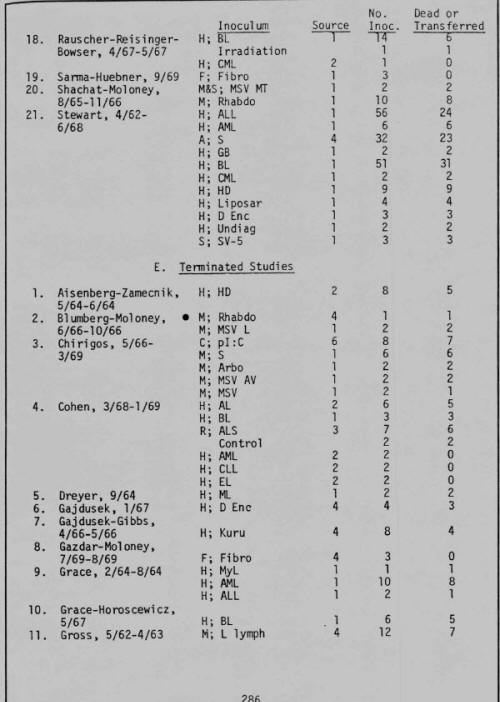
More incredibly, as I turned the pages toward the end of the book, my thumb caught on page 273. There in black and white read "Progress Report on Investigation of Carcinogenesis with Selected Virus Preparations in the Newborn Monkey -- Submitted By Bionetics Research Laboratories, Inc., A Division of Litton Industries." In it was a summary of "The Inoculation Program," wherein between 1962 and 1971, "a total of 2,274 primates have been inoculated" on behalf of "over 70 investigators in 50 different laboratories throughout the world." Here was documented evidence that Litton Bionetics had not only been involved in the development of AIDS-like, Marburg-like, and Ebola-like viruses, but had been inoculating simian monkeys with these mutant horrors as early as 1962, and had shipped the infected monkeys dead and alive, whole and in parts, to labs around the world -- including the ones in which the Marburg and Reston outbreaks occurred.2
Without further ado, I raced back upstairs to photocopy what on gross examination appeared to be the essential parts of the DHEW publication. Then, I did the same with its shelf-mate "Progress Report #9."3 After about an hour of photocopying, I found a chair, sat back, and began to read the volumes from cover to cover. The task ultimately consumed the remaining part of my stay at UNC.
The Special Virus Cancer Program
The Special Virus Cancer Program was introduced by the Program Staff of the Viral Oncology, Etiology Area of the NCI. The finishing touches to the manuscript were added October 24 through 27, 1971, at "the Annual Joint Working Conference, SVCP at the Hershey Medical Center, Hershey, PA.," a location half-way between Bethesda and New York City, and at the same time, I noted, was fairly close to West Point -- Merck-town, U.S.A.
The NCI staff explained:
The Viral Oncology Area is responsible for planning and conducting the Institute's program of coordinated research on viruses as etiological agents of cancer. Scientists within this Area not only provide the broad operational management for intramural and collaborative research but also conduct comprehensive investigations on specific animal oncogenic viruses and their interaction with host cells and apply this information to search for viruses which may be etiologically related to the initiation and continuation of human cancer.
Contract supported research is conducted within the Viral Oncology Program under the Special Virus Cancer Program (SVCP) whose primary objectives are: (I) to determine whether viruses comparable to those known to induce cancers of laboratory and domestic animals are causative agents of human cancer, and (2) to develop therapeutic and preventive measures for control of human cancers when such causative agents are found. A detailed history of events leading to the development of the SVCP may be found in previous Annual Reports of the NCI.
Briefly, in 1964, the Congress of the United States provided funds to the NCI for an intensified program in virus-leukemia research because many scientists were convinced that an effort to identify viruses or to detect virus expression in human tumors would contribute to the determination of the etiology of cancer. Using a new planning approach (Convergence Technique), an overall program aimed on the premise that one virus is an indispensable element for the induction (directly or indirectly) of at least one kind of human cancer and that the virus or viral genome persists in the diseased individual. In 1968, the program was enlarged to encompass all types of cancer. The Program plan is reviewed regularly by the Director, NIH; the Director, NCI; the National Cancer Advisory Council; the Scientific Directorate, NCI; and the Etiology Program Management Group, NCI.
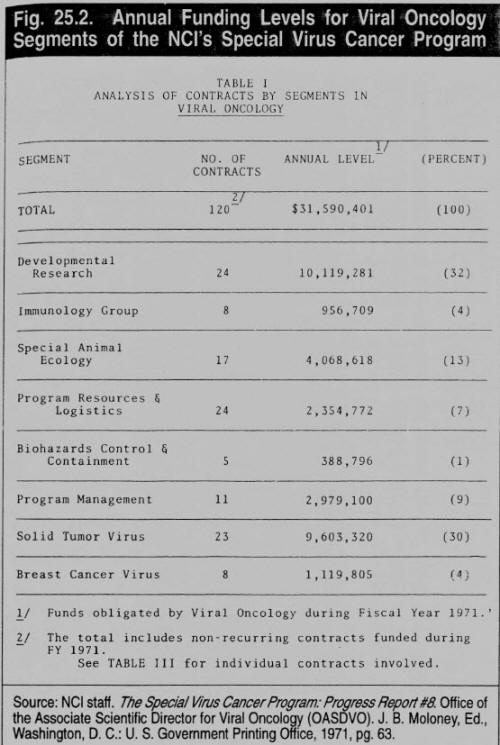
During the past seven years, the Institute has developed an effective management program which has made rapid, substantial progress in understanding cancer induction by viruses. The funding level for this Program in fiscal year 1971 has been about $35 million.4
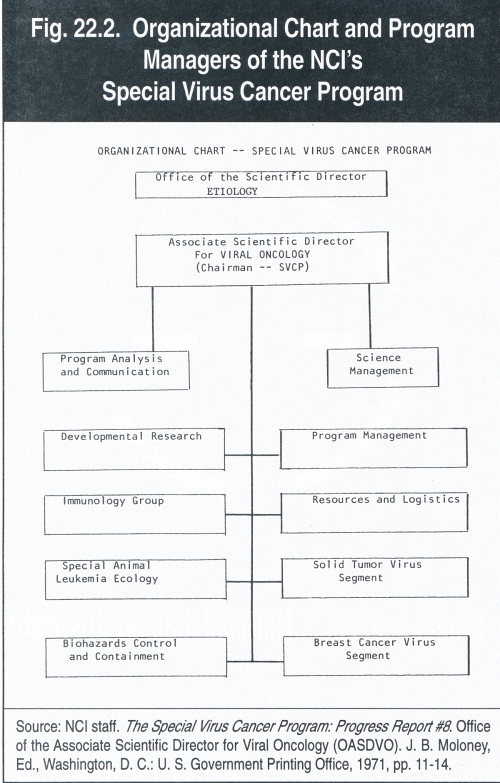
The staff proceeded through a lengthy description of the SVCP's organization and management, which they summarized graphically in an organizational chart and list of managers (see fig. 22.2). This was followed by a list of 105 "Consultants to the Special Virus Cancer Program -- Fiscal Year 1971." Several doctors whose names I immediately recognized were: David Baltimore, MIT; Mark Chatigny, Naval Biological Laboratories; Leon Dmochowski, M.D., Anderson Hospital and Tumor Institute; Peter Duesberg, University of California, Berkeley; Sidney Farber, Children's Cancer Research Foundation, Boston; Peter Gerone, Fort Detrick, Maryland; Richard Herberling and Seymour Kalter, Southwest Foundation for Research; Maurice Hilleman, Merck Institute for Therapeutic Research; John Landon; Bionetics Research Laboratories; Brian MacMahon, one of my professors at Harvard University; Albert Sabin, Weizman Institute, Rehovot, Israel; and Howard Temin, McArdle Research Laboratory, University of Wisconsin; and last, but not least Erling Jensen of Hazleton Research Laboratories. 5
Fig. 22.1. Special Virus Cancer Program Book Cover
This book was removed by NCI administrators from the Fort Detrick library's catalog. It was discovered by the author serendipitously in the basement of the University of North Carolina Davis Library.
Fig. 22.2. Organizational Chart and Program Managers of the NCI's Special Virus Cancer Program
Office of the Scientific Director: ETIOLOGY
Associate Scientific Director For VIRAL ONCOLOGY (Chairman -- SVCP)
Program Analysis and Communication
Developmental Research
Immunology Group
Special Animal Leukemia Ecology
Biohazards Control and Containment
Science Management
Program Management
Resources and Logistics
Solid Tumor Virus Segment
Breast Cancer Virus Segment
Source: NCI staff. The Special Virus Cancer Program: Progress Report #8. Office of the Associate Scientific Director for Viral Oncology (OASDVO). J. B. Moloney, Ed., Washington, D. C.: U. S. Government Printing Office, 1971, pp. 11-14.
***
PROGRAM MANAGEMENT PERSONNEL
Science Management Team
Dr. J. B. Moloney, Associate Scientific Director for Viral Oncology, NCI
Mr. L. R. Carrese, Deputy Associate Director for Program, NCI
Dr. L. R. Sibal, Associate for Program Coordination, Viral Oncology, NCI
Dr. D. J. Rubin, Scientific Coordinator for Extramural Research, VO, NCI
Administrative Officer, Assistant Administrative Officers and Contract Specialists
Mr. John P. Patterson
Mr. Nick Olimpio
Mr. Robert Velthuis
Mr. J. Thomas Lewin
Mr. Maurice Fortin
Mr. Thomas Porter
Mr. Fred Shaw
Mr. John Gibbons
Program Segments and Membership
Developmental Research Segment
Dr. Robert Manaker, Chairman
Dr. Jack Gruber, Vice Chairman and Executive Secretary
Dr. Samuel Dales, Public Health Res Inst., N.Y.C.
Dr. Paul Gerber, DBS, NIH
Dr. Anthony Girardi, Wistar Institute
Dr. Timothy O'Connor, NCI
Dr. Alan Rabson, NCI
Dr. Bernard Roizman, University of Chicago
Dr. Duard Walker, University of Wisconsin
Dr. Virginia Dunkel, NCI
Special Animal Leukemia Ecology Segment
Dr. Michael Chirigos, Chairman
Dr. John Glynn, Vice Chairman
Dr. George Burton, Executive Secretary
Dr. Friedrich Deinhardt, Presbyterian St. Luke's Hospital
Dr. Bernice Eddy, DBS, NTH
Dr. Charles Rickard, Cornell University
Dr. William Moloney, Peter Bent Brigham Hospital
Dr. Mearl Stanton, NCI
Dr. Peter Vogt, University of Washington
Dr. David Yohn, Ohio State University
Dr. Hans Sjogren, Pacific Northwest Research Foundation
Solid Tumor Virus Segment
Dr. Robert J. Huebner, Chairman
Dr. James Duff, Vice Chairman
Mrs. Harriet Striecher, Executive Secretary
Dr. Charles Boone, NCI
Dr. Maurice Green, St. Louis University
Dr. Leonard Hayflick, Stanford University
Dr. Karl Hellstrom, University of Washington
Dr. Edwin Lennette, Calif. Dept. of Public Health
Dr. Joseph Melnick, Baylor University
Dr. Herbert Rapp, NCI
Dr. Wallace Rowe, NIAID, NIH
Dr. Hans Meier, Jackson Laboratories
Immunology Group
Dr. Paul Levine Co-Chairman
Dr. Herbert Rapp
Dr. Ernest Plata, Executive Secretary
Dr. Charles Boone, NCI
Dr. Berton Zbar, NCI
Dr. Tibor Borsos, NCI
Dr. Ronald Herberman, NCI
Dr. Maurice Hilleman, Merck Institute
Dr. Barry Bloom, Einstein College of Medicine
Dr. Arthur Brown, University of Tennessee
Dr. George Santos, Johns Hopkins University
Resources and Logistics Segment
Dr. Robert Holdenried, Chairman
Dr. George Todaro, Vice Chairman
Dr. Roy Kinard, Executive Secretary
Dr. James Duff, NCI
Dr. K. Arnold Fowler, NCI
Dr. Adi Gazdar, NCI
Dr. Paul Levine, NCI
Miss Marie Purdy, NCI
Biohazards Control and Containment Segment
Dr. Alfred Hellman, Chairman
Mr. Errmitt Barkley, Vice Chairman
Mr. Mark Chatigny, Naval Biological Laboratory
Dr. Peter Gerone, Fort Detrick
Dr. Seymour Kalter, Southwest Foundation
Dr. Maurice Mufson, Hektoen Institute for Medical Research
Dr. William Payne, Div. of Environmental Health Sciences, NIH
Dr. Briggs Phillips, Becton-Dickinson
Dr. Arnold Wedum, Fort Detrick
Dr. Richard Griesemer, Ohio State University
Dr. Simon Sulkin, U. of Texas Southwestern Medical School
Dr. George Michaelsen, University of Minnesota
Human Leukemia Therapy Segment
Dr. Seymour Perry, Chairman
Dr. Edward Henderson, Vice Chairman
Breast Cancer Virus Studies Segment
Dr. W. Ray Bryan, Chairman
Dr. Robert Depue, Vice Chairman
Dr. H. J. Clausen, Executive Secretary
Dr. Louis R. Sibal, NCI
Dr. Ernest Plata, NCI
Dr. Harish Chopra, NCI
Dr. J. R. Fraumeni, NCI
Dr. Richard Bates, NCI
Dr. E. Vollmer, NCI
Dr. M. Black, New York Medical College, N.Y.C.
Dr. M. Brennan, Michigan Cancer Foundation
Dr. K. DeOme, University of California, Berkeley
Dr. W. Feller, Georgetown University
Dr. R. Gilden, Flow Laboratories
Dr. D. Moore, Institute for Medical Research
Program Management Segment
Dr. J. B. Moloney, Chairman
Dr. L. R. Sibal, Executive Secretary
Dr. W. Ray Bryan, NCI
Dr. A. J. Dalton, NCI
Dr. James Duff, NCI
Dr. Michael Chirigos, NCI
Dr. Robert Huebner, NCI
Dr. Robert Manaker, NCI
Dr. Deward Waggoner, NCI
Dr. Robert Holdenried, NCI
Dr. George Todaro, NCI
SVCP Highlights
Next, "Progress Highlights" of their scientific activities were presented. The majority of highlights focused on Type C viruses which, of course, Gallo's group had manipulated by the dozens. The NCI staff reported "There are now over 100 viruses which are known to cause virtually all kinds of cancer in every major group of animals including nonhuman primates."
Viruses of one (Type C) are known to cause leukemias, lymphomas, and sarcomas in chickens, mice, and cats. Particles, which closely resemble Type C viruses, can be found in human patients affected with these same kinds of cancers.
. . . By using techniques developed from animal tumor virus studies, every effort is being made to determine whether these viruses cause human malignancies .... Some important new discoveries that have been made are: (1) Certain oncogenic viruses are unable to produce malignancies unless a "helper" virus is present [such as EBV], thus suggesting that an interplay exists between two viruses. (2) Certain tumor-inducing chemicals, irradiation, (carcinogens) may act as cofactors in activating latent, oncogenic viruses within cells. [This dovetailed what Peter Duesberg had argued with regard to AIDS.6] (3) Certain tumor viruses contain unique enzymes which are required in the replication of viruses in cells [exactly what Gallo and coworkers from Litton Bionetics had been studying with the reverse transcriptase enzyme]. These and other important developments will be discussed further in this report.5
Type C Particles
When the Special Virus Cancer Program was initiated, highest priority was given to the search for human leukemia viruses resembling the Type C viruses causing chicken and mouse leukemias. Since that time, many Type C viruses, the total is now about 85, have been found in a variety of tumors from many species of two vertebrate classes. All of these species continue to be studied intensively under the broader scope of the Special Virus Cancer Program. Several of the Type C viruses are established as the causative agents in leukemias, lymphomas, and sarcomas of chickens, mice, cats and hamsters. Many of these can infect and produce malignancies in other species (e.g. a sarcoma virus of the cat produces tumors in marmoset monkeys). Furthermore, some of these viruses can cause malignant transformation to occur in animal and human cells grown in the laboratory (e.g. cat leukemia and sarcoma viruses alter embryonic human cells). Type C virus particles have been found in association with malignancies of a spectrum of animal species including nonhuman primates, rats, cattle, woolley monkeys, gibbons, and man .... 5
After reading this, I reflected on "Gallo's Research Anthology" (see fig. 6.8). I recalled he not only received credit for discovering the first two types of human leukemia viruses, HTLV-I and II, but he published manipulating Type C monkey viruses-he extracted the viruses' nucleic acids, infused their empty envelopes with chicken and cat leukemia/sarcoma RNA, grew the mutants in human WBC cultures, then prompted them to infect human T-Iymphocytes and produce the leukemias, sarcomas, general wasting, and essentially all the symptoms of AIDS (see fig. 6.5).7
Creating More AIDS-like Viruses
Under the report's next heading, "Reactions between Type C viruses causing leukemias and sarcomas (solid tumors)," the NCI staff explained how and why they created more AIDS-like viruses:
When inoculated into appropriate cell cultures, Type C sarcoma viruses of chickens, mice and cats produce foci [cancerous growths] of altered cells. This fundamental discovery provides a readily visible indicator reaction for the detection of sarcoma viruses. On the other hand, leukemia viruses grown in tissue culture do not cause foci or other detectable changes. The finding that leukemia viruses can either inhibit or enhance focus formation by sarcoma viruses of the same species has led to the development of methods for the detection and quantitation of leukemia viruses indirectly.
Certain of the chicken, cat and mouse sarcoma viruses are "defective" in that they do not produce foci in cell cultures or tumors in animals in the absence of a co-infecting, "helper" leukemia virus. [Amazingly, the researchers called carcinogenic viruses "defective" if they were unable to produce cancers without the help of other factors including chemicals, radiation, and here leukemia viruses.] Further, in the presence of a defective sarcoma virus the helper action of leukemia viruses can be used as a specific indicator for their detection and quantitation. It is now believed that defective sarcoma virus-leukemia virus interactions may be more widespread in nature than originally thought and that similar systems may be found in man. A mouse leukemia virus which has been adapted to grow in human cells is now available to search for defective human sarcoma viruses, if they exist.8
In other words, humans would eventually be inoculated with mouse leukemia viruses in an effort to locate inactive human sarcoma viruses. Like the BLV experiments Strecker described in cows, here humans were to become the "mixers" for mutant leukemia and sarcoma viruses.
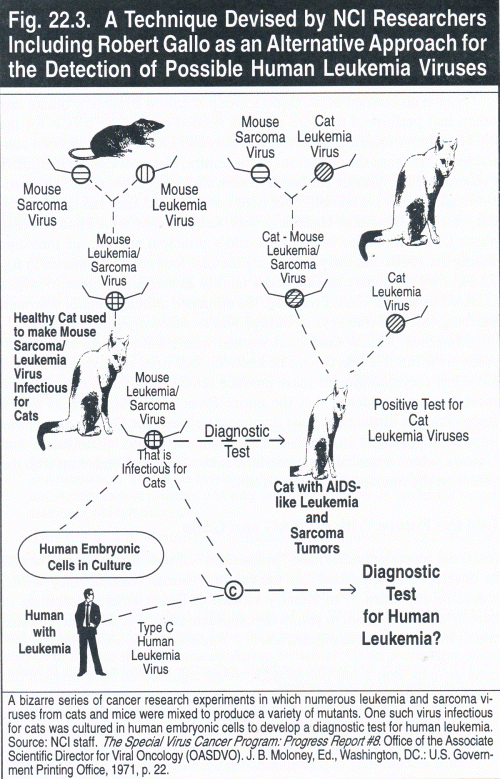
Here, in almost lay terms, the NCI staff described their greatest accomplishment in 1971 was to produce AIDS-like viruses for pre-cancer diagnosis. In continuing this effort, they reported an "alternative approach" had been developed for the detection of possible human leukemia viruses:
A defective mouse sarcoma virus and its leukemia virus helper can be made to form tight functional aggregates, which behave as one virus. Using a mixture of mouse sarcoma virus and cat leukemia virus, a hybrid aggregate which could be grown continuously in cat cells was produced. [Again, this is exactly what Gallo reported.7] Because the aggregate is defective, it requires the simultaneous presence of a cat leukemia virus for producing altered foci in cat cells. Thus, a focus forming sarcoma virus of the mouse, artificially changed to one possessing infectivity for cat cells, can now be used in cultures for the detection of cat leukemia viruses.
This hybrid virus, as well as the cat leukemia virus, will also grow in human embryonic cells in tissue culture. If sufficient amounts of the Type C particles found in association with human leukemia can be obtained, the possibility exists that the cat-adapted mouse sarcoma virus can be hybridized with the human agent to produce an indicator system for the detection of human leukemia viruses.8 Fig. 22.3 presents a graphic description of this work.
Litton's Group in Uganda
The NCI staff went on to explain how Type C virus particles can get into the genes of normal cells and cause them to develop cancer. Then they discussed in great detail, the enzyme that represented "a dramatic breakthrough" in the investigation of cancer. Without naming Gallo, they discussed his explicit area of expertise -- RNA-dependent DNA polymerase -- and explained, as Gallo had in numerous publications, the "biochemical pathways of tumor virus infection and replication."
Intensive investigations have now revealed polymerase activity in cells of patients with acute lymphoblastic leukemia; more preliminary evidence has shown the enzyme is in cells of sarcomas, Burkitt's lymphoma and breast cancer. Since the RNA-dependent DNA polymerase is apparently always present in the RNA tumor viruses of animals, its discovery in the human tumor cells offers good supportive evidence that viruses are associated with cancers in man. The RNA-dependent DNA polymerase of human leukemia cells is inhibited by a drug, n-dementhyl rifampicin [which Gallo had received from another documented Nazi employer and biological weapons contractor, Dow Chemical Company, and which he subsequently studied and reported on,9 and] which also inhibits the enzyme activity found in the Type C RNA tumor viruses of animals. Studies are underway [principally in Gallo's lab] to explore the action of this drug and the various modifications of it. These investigations could provide new approaches to the treatment of malignancies in man.8
Fig. 22.3. A Technique Devised by NCI Researchers . Including Robert Gallo as an Alternative Approach for the Detection of Possible Human Leukemia Viruses
A bizarre series of cancer research experiments in which numerous leukemia and sarcoma viruses from cats and mice were mixed to produce a variety of mutants, One such virus infectious for cats was cultured in human embryonic cells to develop a diagnostic test for human leukemia. Source: NCI staff. The Special Virus Cancer Program: Progress Report #8. Office of the Associate Scientific Director for Viral Oncology (OASDVO). J.B. Moloney, Ed., Washington, DC.: U.S. Government Printing Office, 1971, p. 22.
Following a shorter review of Type B viruses thought to be associated with breast cancer, and Herpes-type viruses "associated with some forms of chronic leukemia, lymphoma and postnasal carcinoma ... " the NCI staff focused on EBV. Their program, they said, had studied the significance of Epstein-Barr viruses extracted from Burkitt's lymphomas and postnasal carcinomas "through the International Agency for Research on Cancer (IARC) in the West Nile District of Uganda." This, of course, was close to where Preston noted AIDS, Marburg, Ebola, and Reston viruses were believed to have originated. The purpose of the NCI/NARC collaborative effort was "to determine the feasibility of further studies on EBV in relation to Burkitt's tumor .... " Also mentioned was the fact that "EBV infection has been associated with the development of infectious mononucleosis in young adults, a disease with the attributes of a self-limiting leukemia."10
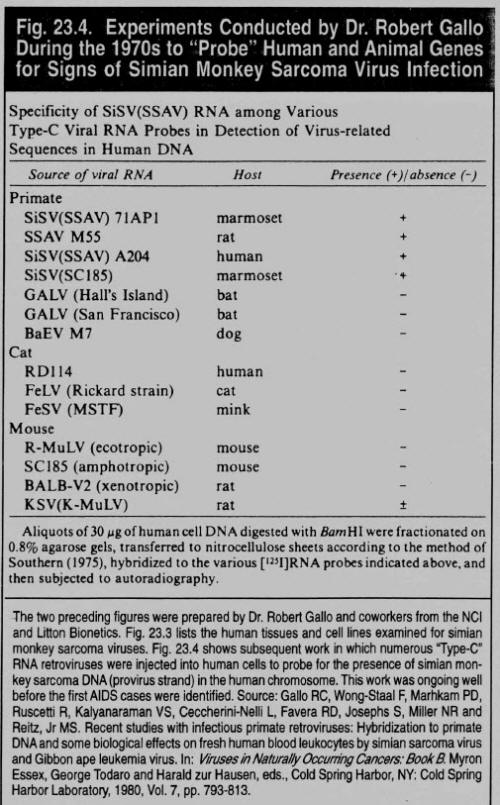
Now it all seemed to fit. Gallo's group at Litton Bionetics, unlike any other, had performed the core services needed during the SVCP for the NCI to achieve its principle goals. Not only had Litton Bionetics been contracted to "manage the operations of the [entire] National Cancer Institute's Frederick (Md.) Cancer Research Center, including more than 750 scientists and support personnel," I I but here was evidence they ran the show in the West Nile District of Uganda as well. Reflecting on what I had just read about Litton Bionetics being the world's principal supplier of monkeys during the 1960s and early 1970s (see the first four grant summaries in fig. 22.5),2 they obviously maintained a facility in this area -- close to where OTRAG operated. Thus, I realized, the unnamed animal supplier who sent Marburg-infected monkeys to Europe was undoubtedly Litton Bionetics.
Moreover, I knew Gallo had worked with EBV to make his retroviruses grow faster. I was certain he knew his cell lines needed to be free of EBV lest they continue to mass produce retroviruses. 12 But here was the most damning evidence that the entire French-American fracas was a farce -- truly a distraction. This document proved that Gallo's group was intimately involved in EBV studies simultaneously in Bethesda and Uganda, which would also explain how Litton Bionetics ended up with the patent on the mononucleosis blood test. 13
Into the Future With OTRAG and Gallo
Next, the report provided their "projections" of how their research would be developed or expanded into the future. "Human studies," they wrote, would be expanded "to identify viruses or detect virus expressions in human tumors ... [and] to search for candidate viruses or subviral products which induce human malignancies .... " Such studies were desired:
(1) To identify and isolate candidate viruses or subviral products in leukemias, lymphomas and sarcomas.
(2) To identify and isolate candidate viruses or subviral products in breast, lung and other carcinomas.
(3) To develop methods for the detection of high cancer risk groups, i.e., individual susceptibility of predisposition to transformation by human viruses.
(4) To develop suitable reagents for mass diagnostic screening for candidate viruses.
(5) To characterize, biologically and biochemically, presumptive viral agents.
(6) To increase emphasis on understanding the relationship of environmental agents (e.g. chemical carcinogens) as cofactors in viral carcinogenesis. This represents a major expansion of effort requiring combined efforts of the Viral Oncology and Chemical Carcinogenesis Areas. 14
This later function was one Litton Bionetics's chief revenue producing specialties.
The SVCP's projections regarding future "Molecular Studies," which again was Dr. Gallo's forte, and their "Immunological Studies" in which Hilleman, Krugman, and Poeisz majored were summarized next (see fig. 22.4).
Next, the SVCP "Summary Reports" reviewed progress which had been made in every working group. In this section, the staff disclosed the principal researchers directing each group and area of investigation.
Under another heading "Other Research Developments" in the Viral Leukemia and Lymphoma Branch, the staff described a variety of viral experiments, including those using Epstein-Barr, cat leukemia/sarcoma, and monkey leukemia/lymphoma/sarcoma viruses. Here they announced a major breakthrough:
Herpes saimiri is a DNA containing virus indigenous to the squirrel monkey. Recent studies have shown that this virus will induce acute lymphocytic leukemias as well as lymphomas and reticulum cell sarcomas in owl monkeys and marmosets. This is the first demonstration that a virus produces leukemia and lymphomas in primates and may serve as an important model for human leukemia and lymphoma. 15
This disclosure obviously foreshadowed the announcement made almost ten years later by Gallo that he had discovered HTLV-I, a leukemia virus he identified as the cause of T-cell leukemia in Japan.
I thus wondered whether H1LV-I had been developed by Gallo and company during the early 1970s as part of the SVCP. HTLV-I, like HIV and the Marburg viruses, may have then been accidentally released or deployed to initiate the Japanese epidemic for which Gallo could later claim the discovery of its etiologic agent. This would put a new spin on what Shilts described as "something of a backward scientific affair," I realized. 16 In fact, the entire H1LV-I saga, in light of this evidence, was reasonably a possible pilot study or model for the AIDS epidemic a decade later.
Fig. 22.4. A Description of the Molecular and Immunological Studies Conducted During the NCI's Special Virus Cancer Program
Molecular Studies
In recent months rapid major advances have been made in the field of molecular biology. These findings have direct application to the study of the relationship of viruses to tumors. There is evidence that the genetic material (RNA) of the tumor viruses can direct the synthesis of new DNA. The demonstration that RNA tumor viruses contain enzymes (polymerase, ligase) which may be required for viral infection, interaction with host cell genome, and viral replication has provided the basis for the development of new, extremely sensitive methods for the detection of oncogenic viruses or their "fingerprints." Indeed, knowledge of the fundamental molecular events which occur during virus infection and subsequent cell transformation provides the first truly rational approach to therapy. Enzyme activities analogous to those of RNA tumor viruses have recently been found in cells of human leukemics. This offers strong supportive evidence that viruses are associated with cancers in man.
a. Basic studies
The Program is prepared to broaden its activities for identifying and characterizing the spectrum of enzymes (and other mediators) required by tumor viruses for replication and transformation.
b. Applied studies
As knowledge of the fundamental molecular events in virus-cell interactions is developed, the Program will apply this information to the study of human cancer as follows:
(1) To identify and characterize similar enzymes or enzymatic activities within normal and malignant human cells.
(2) To develop highly sensitive methods for the detection of virus or virus activity in human cells.
(3) To develop a rational basis for therapy or prevention by exploring various approaches to blocking of viral replication and/or tumorigenesis at the cellular and subcellular levels. The therapy could be directed at any or all of the stages of cell transformation beginning with cell infection by a tumor virus.
Ultimately these studies will require an exhaustive effort to develop drugs, anti-enzymes, gene repressors or inhibitors effective at the molecular level.
3. Immunological Studies
Immunologic research has provided extremely sensitive techniques for detection and characterization of tumor viruses, viral antigens, and changes in surface membranes of tumor cells. Indeed, such efforts have contributed to an understanding of the role of immunological mechanisms in host-tumor and host-virus interactions which provide an approach to the prevention and treatment of cancer.
a. Basic studies. Investigations of selected model systems, representing tumors induced by Type C, Type B, and Herpes-type viruses, will be extended to further identify, characterize and determine the viruses, viral antigens, and membrane antigens of tumor cells. This includes development and application of improved techniques with the sensitivity and specificity required to detect cellular alterations induced by tumor viruses alone or as the result of interaction with other environmental agents (e.g. chemicals, irradiation). Efforts will be increased to develop similar immunological methods and diagnostic reagents for application to human cancer. Research will be intensified and expanded:
(1) To study cellular and humoral immune mechanisms and to determine their relative significance in host recognition of and response to tumor and/or tumor viruses.
(2) To develop methods to enhance host response to tumor or virus antigens.
Increasing emphasis will be directed toward research on spontaneous or naturally occurring tumors in model systems relevant to human cancer. These studies would provide the basis for a rational approach to prevention (vaccines) and treatment (immunotherapy) of cancer.
b. Applied studies. Basic research will provide the framework or identification and characterization of viruses, viral antigens, and cell membrane alterations in human cancers. Immunological methods and reagents will be developed and applied:
(1) To relate candidate human viruses to known oncogenic agents.
(2) To identify and characterize interspecies viral antigens which are present in known mammalian tumors, and therefore, could provide the basis for a formidable probe to detect human tumor viruses or viral antigens.
(3) To launch large-scale seroepidemiological surveys which will define high risk populations.
(4) To determine the presence of cross-reacting antigens in various human tumors.
Clinical studies will be directed toward understanding and manipulation of immune mechanisms in human cancer as a basis for:
(1) Development of vaccines from identified and fully-characterized human tumor virus(es).
(2) Determination of the role of host immune responses in tumor recognition and rejection.
(3) Application of (1) and (2) in the prevention and control of human cancer.
As research progresses, increased emphasis on application will be as follows:
(1) Immunodiagnosis and seroepidemiology
(2) Clinical studies on the role of immune mechanisms in human cancer
(3) Immunotherapy
(4) Vaccines (conventional or other)
Ultimately, these studies would be organized to coordinate and integrate the application of appropriate biochemical, immunological, and genetic methods of detection, prevention, and control of various types of human cancer.
4. Test Systems
In vitro and in vivo (animal) test systems will be carefully selected to evaluate the work outlined in the previous research areas; specifically: (a) to determine the oncogenic potential of candidate human viruses; (b) to develop bioassay systems for testing viral, and viral/chemical carcinogens; (c) to begin vaccine (conventional or other) testing and immunization programs; (d) to begin therapy testing programs; and (e) to explore special animal tumor systems with particular relevance to human cancer.
Source: NCI staff. The Special Virus Cancer Program: Progress Report #8. Office of the Associate Scientific Director for Viral Oncology (OASDVO). J. B. Moloney, Ed., Washington, D. C.: U. S. Government Printing Office, 1971, pp. 28-30.
A Cat Owner's Nightmare
Another paragraph dealt with cat owners and AIDS-like diseases in cats. Having studied feline leukemia/sarcoma viruses, the staff reported, "About one and one-half years ago, a nationwide effort was initiated to obtain cancerous cats (primarily leukemias and sarcomas) from cat breeders and owners." The NCI put out a nationwide call "for materials and cancerous purebred cats with pedigrees." Letters were "sent to cat owners soliciting cats with veterinarian-diagnosed cancers."15
The authors wrote, "The procurement effort not only obtains valuable research material but reassures the cat owners and other concerned citizens of NCI's active program for the evaluation of the unlikely hazard of this disease to human health."15
How ironic, I thought, the NCI was spending hundreds of millions of taxpayer dollars to prove that animals, including cats and monkeys, were potential reservoirs for cancer viruses at the same time they assured people that it was safe to handle these animals. Then, even more bizarre, was the fact that as pet owners supplied the NCI with experimental cat viruses, the researchers engineered them to cause cancer in humans.
Apparently, I realized, cancer virus-infected animals posed less of a risk to the public's health than did the NCI.