Response to Imported Case of Marburg Hemorrhagic Fever, the Netherlandsby Aura Timen, Marion P.G. Koopmans, Ann C.T.M. Vossen, Gerard J.J. van Doornum, Stephan Günther, Franchette van den Berkmortel, Kees M. Verduin, Sabine Dittrich, Petra Emmerich, Albert D.M.E. Osterhaus, Jaap T. van Dissel, and Roel A. Coutinho1
Volume 15, Number 8—August 2009
National Institute for Public Health and the Environment, Bilthoven, the Netherlands (A. Timen, M.P.G. Koopmans, S. Dittrich, R.A Coutinho)
Leiden University Medical Center, Leiden, the Netherlands (M.P.G. Koopmans, A.C.T.M. Vossen, J.T. van Dissel)
University Medical Center, Rotterdam, the Netherlands (M.P.G. Koopmans, G.J.J. van Doornum, A.D.M.E. Osterhaus)
Bernhard-Nocht-Institute for Tropical Medicine, Hamburg, Germany (S. Gunther, P. Emmerich)
Elkerliek Hospital, Helmond, the Netherlands (F. van den Berkmortel, K.M. Verduin)
St. P.A.M.M., Veldhoven, the Netherlands (K.M. Verduin)
Academic Medical Center, Amsterdam, the Netherlands (R.A. Coutinho)
European Centre for Disease Prevention and Control, Stockholm, Sweden (S. Dittrich)
1 On behalf of the national response team.
Address for correspondence: Aura Timen, PO Box 1, 3720 BA Bilthoven, the Netherlands; e-mail:
[email protected]NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHTYOU ARE REQUIRED TO READ
THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
AbstractOn July 10, 2008, Marburg hemorrhagic fever was confirmed in a Dutch patient who had vacationed recently in Uganda. Exposure most likely occurred in the Python Cave (Maramagambo Forest), which harbors bat species that elsewhere in Africa have been found positive for Marburg virus. A multidisciplinary response team was convened to perform a structured risk assessment, perform risk classification of contacts, issue guidelines for follow-up, provide information, and monitor the crisis response. In total, 130 contacts were identified (66 classified as high risk and 64 as low risk) and monitored for 21 days after their last possible exposure. The case raised questions specific to international travel, postexposure prophylaxis for Marburg virus, and laboratory testing of contacts with fever. We present lessons learned and results of the follow-up serosurvey of contacts and focus on factors that prevented overreaction during an event with a high public health impact.
Keywords: Marburg virus diseases, hemorrhagic fever, exposure, contacts, temperature monitoring, filovirus, viruses, perspective
In Western countries, Marburg hemorrhagic fever (MHF) is an imported disease with a low risk of occurrence, but it has a high profile in the public mind (1) because it can be transmitted from person to person, the course is fatal in up to 80% of cases, and the reservoir is uncertain (2,3). The infection is caused by the Marburg virus (MARV), an enveloped, nonsegmented, negative-stranded RNA virus belonging, with the Ebola virus, to the family Filoviridae. Although the main transmission route is direct contact with blood or other infected body fluids, transmission by droplets and aerosols cannot be ruled out and has been demonstrated in animal models (4).
MARV was identified in 1967 in Marburg, Germany, during a laboratory outbreak caused by handling tissues of African green monkeys (5,6). From 1975 through 1987, sporadic cases occurred in South Africa (1975, when the index case, a person exposed in Zimbabwe, was diagnosed in South Africa) (7) and in Kenya (1980, 1987) (8–10). Outbreaks were reported from the Democratic Republic of Congo in 1998–2000 (11,12), Angola in 2004–2005 (2) and Uganda in 2007 (13). Nonhuman primates and bats are suspected as sources of infection, but their role in the natural reservoir for MARV and transmission to humans is unclear (14).
In July 2008, an imported case of MHF was diagnosed in the Netherlands. We describe the public health response involving the management of 130 contacts at risk of acquiring the disease.
The CaseOn July 5, 2008, a 41-year-old woman was referred by her general practitioner to the Elkerliek Hospital because of fever (39°C) and chills of 3 days’ duration after returning from a June 5–28 holiday in Uganda. She was placed in a hospital room with 3 other patients. Malaria was ruled out by 3 negative blood films. Routine bacteriologic tests were performed, and empiric treatment with ceftriaxone, 2 g/day, was started. On July 7, hemorrhagic fever was included among other infectious causes in the differential diagnosis because of rapid clinical deterioration and impending liver failure. An ambulance stripped of all unnecessary devices and equipped in accordance with strict isolation protocols transferred the patient to a single room with negative air pressure ventilation and anteroom in the Leiden University Medical Centre (LUMC).
After admission, rash, conjunctivitis, diarrhea, liver and kidney failure, and finally, hemorrhaging developed in the patient. Extensive bacteriologic and virologic analyses were conducted, and plasma samples were sent to Dutch national laboratories and to the Bernhard-Nocht-Institute for Tropical Medicine (BNI) in Hamburg, Germany, for testing to detect antibodies to and RNA from filoviruses. Initial laboratory results from the Dutch national reference laboratory were ambiguous for hemorrhagic fever. On July 10, BNI reported a positive reverse transcription–PCR result for MARV (15), which was confirmed by sequence analysis of the polymerase gene. The strain was related to, but distinct from, known isolates. MARV was confirmed by PCR by the Department of Virology at Erasmus Medical College (Rotterdam, the Netherlands). On July 11, the patient died of consequences of cerebral edema.
Travel History and Hypotheses for the Source of InfectionThe patient’s travel group consisted of 7 Dutch tourists and 2 guides. Three of the tourists, including the patient, and 1 guide visited an empty cave on June 16 in Fort Portal and the Python Cave in the Maramagambo Forest on June 19. The patient’s partner recalled bats flying around in the latter cave, bumping against the visitors, and large amounts of droppings on the ground. She incurred no bite wounds, and no preexisting wounds were exposed to bats. On July 23, the travel group came within 5 m of gorillas in the wild and visited a village inhabited by pygmies, where they saw an elderly sick woman lying under a blanket.
We postulated that the most probable source of MARV infection was the visit to the Python Cave, known for its colony of Egyptian fruit-eating bats
(Rousettus aegyptiacus). The party had photographed these bats, and this species of bat has been shown to carry filoviruses, including MARV (16,17) in other sub-Saharan locations. We estimated the incubation period of the infection to be 13 days.
Organization of Public Health ResponseOn July 8, the attending physician at the LUMC notified the Dutch public health authorities about the case. A national outbreak response team was formed of clinicians, medical microbiologists and virologists, public health specialists, staff members from the national response unit, and a press officer. This team convened a nearly daily teleconference to 1) to perform a structured assessment of the public health risks in the 2 hospitals and in the community, 2) perform risk classification of contacts, 3) issue guidelines for follow-up, 4) provide information to professionals and media, and 5) monitor progression of crisis response.
Immediately after the diagnosis was confirmed, on July 10, a press conference was held. Various press statements emphasizing the control measures designed to prevent secondary transmission followed the press conference. The World Health Organization was notified according to the International Health Regulations by the National Focal Point, and international warnings were issued through the Early Warning and Response System and through ProMED.
Management of ContactsAlthough MARV infectivity is highest in the last stage of the disease, when severe bleeding coincides with high viral load, we considered the onset of fever (July 2) as the starting point for contact monitoring. Follow-up measures tailored to the risk group were undertaken during the 21 days after last possible exposure (14,18,19). The high-risk group comprised anyone with unprotected exposure of skin or mucosa to blood or other body fluids of the index patient. It included the other 3 patients in the patient’s room at Elkerliek and personnel who handled her specimens without protection. The low-risk contacts were LUMC and ambulance personnel who had employed the appropriate personal protective measures while caring for the patient or diagnostic samples. Persons who had been near the patient during her holiday, the return flight, and stay in the Netherlands until Elkerliek admission but who were not exposed to her body fluids during her febrile illness and personnel from reference laboratories who worked under BioSafety Level 3 conditions were categorized as casual contacts.
A total of 130 at-risk contacts were identified, 64 at high risk and 66 at low risk (Table). High-risk contacts were required to record their temperature 2×/day, report to the local health authorities 1×/day, and postpone any travel abroad. The low-risk contacts were asked to record their temperature 2×/day and to report to local health authorities if it was>38°C. No limits were imposed on the casual contacts.
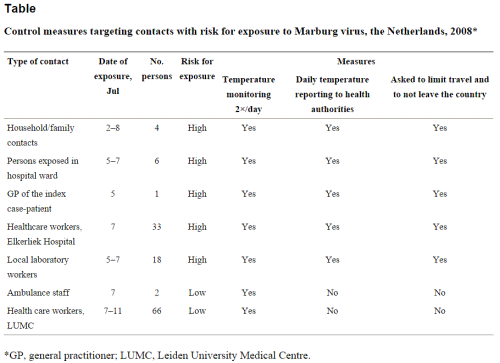 Control measures targeting contacts with risk for exposure to Marburg virus, the Netherlands, 2008*
Control measures targeting contacts with risk for exposure to Marburg virus, the Netherlands, 2008*Because asymptomatic MARV infection is rare (20,21) and thus unlikely to play a role in spreading the infection, we restricted further clinical and laboratory evaluation to persons with a temperature >38°C, measured at 2 points 12 hours apart. Every case of fever was to be assessed on an individual basis by the response team. Three academic hospitals provided stand-by isolation facilities for admission of contacts.
On August 1, the temperature monitoring of contacts ended. Fever of at least 12 hours’ duration or clinical signs of MHF did not develop in any of the contacts. Fever within 21 days did not develop in any of the travel companions and local guide who joined the patient in the bat cave. Because sustained fever did not develop in any of the high-risk or low-risk contacts during the surveillance period, no clinical or laboratory follow-up for MARV was needed. The Technical Appendix summarizes other findings during the monitoring period, dilemmas encountered with respect to travel restrictions, postexposure options in case of a high-risk accident, and laboratory diagnosis in the early stage of infection. The Technical Appendix also describes laboratory procedures used.
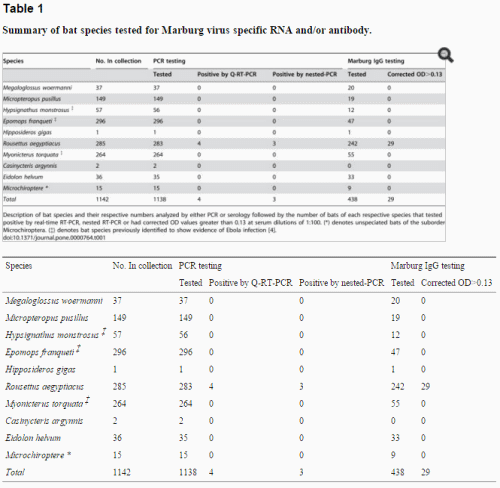
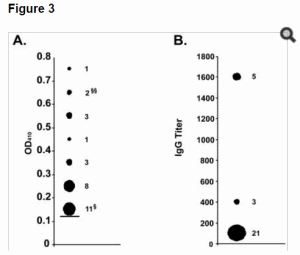
Serologic Follow-upTo identify asymptomatic seroconversion, a serosurvey was undertaken of 85/130 (65%) contact persons who participated in the study. They represented 78% (50/64) of high-risk contacts and 53% (35/66) of low-risk contacts and included the Dutch visitors to the bat cave. Blood samples were collected from December 2008 through February 2009, 5–7 months after possible exposure. The laboratory testing was performed at the BNI in Hamburg by using an immunofluorescent antibody (IFA) assay.
The IFA slides were prepared using the MARV strain of the index patient. Details about the laboratory testing are given in the Technical Appendix. In 2 initial evaluations, all but 2 samples were negative for antibodies against MARV. Additional screening found that all serum samples tested negative for immunoglobulin (Ig) G and IgM to MARV.
DiscussionWe have described the public health response to the case of MHF in a Dutch woman returning from travel abroad, who was most likely exposed to MARV by visiting a bat cave. Outbreaks caused by filoviruses constitute a serious public health threat in sub-Saharan countries and have disruptive consequences at the individual and societal level. In countries in which these viruses are not endemic, imported cases occur only sporadically and are associated with little or no secondary transmission (22). Our patient represents a rare case of MARV infection imported to a Western country, and her case is unusual in that her only likely exposure was visiting a bat cave while traveling in Uganda. Insectivorous bats may have been the source of sporadic cases in Zimbabwe in 1975 (23) and Kenya in 1980 and 1987 (8,9). Furthermore, epidemiologic evidence linked a large outbreak of MHF in Durba (Democratic Republic of Congo) to a mine containing a large population of fruit-eating bats (24). Although the source of infection in our case is not certain, circumstantial evidence points to transmission in the Python Cave. Ecological surveys to assess the presence of infected bats in that cave are ongoing (P. Rollin, pers. comm.).
Our case shows that unnoticed exposure to an unknown reservoir in a country with no apparent cases of MHF can lead to infection. In countries with previous cases of MHF, entry into bat caves should certainly be avoided until we know the role of bats as reservoir for MARV. The importance of MHF for western countries may be increasing, with more persons traveling to high-risk regions and incurring exposure by intrusion into unaccustomed ecological niches. Hospital staff in low-risk countries must be alert to this possibility. In most travelers returning from tropical destinations, fevers are caused by common pathogens or by malaria. However, fever together with rapid clinical deterioration and hemorrhaging in a patient returned from a suspect region should suggest viral hemorrhagic fevers, especially if exposure to a possible reservoir could have occurred.
Inclusion of MHF in the differential diagnosis of a patient triggers an immediate public health response. This response aims primarily at reducing the chance of secondary transmission by identifying contact persons at risk. Person-to-person transmission occurs in countries to which MARV is endemic (22) but only once has been reported elsewhere (23). In this case we identified 130 contacts with possible risk. Two hospitals, 2 public health departments, and 3 laboratories were involved. We decided to trace all people who were in contact with the index patient after her fever developed and to assess their risk for exposure on a case-by-case basis. All contacts complied with temperature monitoring and daily reporting. All but 2 high-risk contacts postponed further travel until the theoretical incubation period of 21 days had elapsed.
In the Netherlands, statutory power to prevent a healthy person from traveling abroad is limited, but the Public Health Law is being revised, and emergency legal provisions are being considered. Despite various recommendations (14,18,25–27), no evidence-based, widely accepted international protocol is available to guide contact classification and monitoring in the case of MHF. Legislation on containment of dangerous pathogens (1) and measures applied to contacts differ among countries, sometimes with extreme consequences. These differences, together with privacy issues, make international exchange of information difficult.
The serosurvey of the contacts of this patient confirm that no secondary transmission took place between her and any contact who provided a blood sample. Our results are consistent with those of Borchert et al. (21), who found no serologic evidence for asymptomatic or mild MARV infection in a serosurvey of household contacts.
The present case was an exceptional situation in which visiting a tourist attraction led to MHF, a disease with a high potential for overreaction. Given this potential, a rational response must be built on a thorough and evidence-based risk assessment (1). The response in the Netherlands was low profile and did not lead to overreacting or public alarm. Its key factors were a coordinated risk assessment and contact monitoring, together with factual updates for health professionals and the public. MHF may be more often encountered in industrialized countries in the future due to adventure travel to regions endemic for MHF.
Supplementary Material
Technical Appendix:
Response to Imported Case of Marburg Hemorrhagic Fever, the Netherlands
Technical Appendix
Clinical Findings and Dilemmas during Contact Monitoring
Clinical Findings
In 1 high-risk contact, the body temperature once exceeded 38°C, but 12 hours later, the temperature had normalized. Another high-risk contact who shared the patient’s room at Elkerliek Hospital was readmitted to that hospital because of heart failure, pulmonary congestion, and subfebrile temperature. His first admission had ended days before his readmission; his condition did not differ between stays and could be attributed to the underlying end-stage heart disease. Several other contacts showed nonspecific symptoms such as nausea and headache, but without fever, and specific follow-up was deemed unnecessary.
The monitoring period led to emotional problems, mostly in high-risk contacts, due mainly to the restrictive measures on daily life and the relatively long period of uncertainty about their prognosis and possible transmission to family members. Psychological support was made available on a case-by-case basis by the occupational health department of the 2 hospitals.
Dilemmas in the Management of Contacts
Problems arose regarding international travel, testing of contacts, and postexposure prophylaxis. By the time Marburg hemorrhagic fever was diagnosed in the index patient, 2 contacts had left for holidays in Italy and the United States, respectively, where they remained for most of the monitoring period. The national authorities of both countries were contacted, and the protocols for temperature monitoring were conveyed with follow-up information on the health status of the 2 persons.
A third contact departed for Poland 3 days before completing the monitoring, after being instructed to carry on the monitoring and stay in daily contact with the Dutch authorities. The Polish authorities were informed because there were doubts about his compliance. Another person left for Morocco 1 day before the end of the monitoring period, but he kept in touch with the Dutch authorities.
To anticipate possible needlestick accidents or gross breaches of isolation measures by healthcare workers, use of experimental vaccines were assessed in a teleconference with international experts. They favored the vaccine in which attenuated recombinant vesicular stomatitis virus vector expresses the Marburg virus (MARV) glycoprotein (1–3) and developed protocols for its use, including regulatory aspects and measures to contain environmental shedding of VSV.
Laboratory Diagnosis in the Early Stage of Infection
Transportation and Processing of Samples
Transport of samples must be organized before sample collection to avoid bottlenecks. We therefore arranged for certified couriers to link hospitals quarantine facilities to laboratories, including the nearest reference laboratory in Germany.
Protocols were designed to encompass essential laboratory testing of severely ill patients, including chemical and bacteriologic diagnostic techniques, biosafety considerations, and methods for decontamination of equipment. No existing preparedness protocols included these considerations. We decided that diagnostic work-ups would be limited to contacts in whom fever developed. In that case, essential equipment for blood chemistry analyses would be placed inside the Intensive Care isolation facility.
Laboratory Assessment of Febrile Contacts: Differential Diagnosis
Protocols were developed for diagnosis of the most probable causes of illness, given the seasonal patters, in which prodromal symptoms resemble those seen in patients with a filovirus infection. These include fever, myalgia, and diarrhoea. Data from physician-based studies of respiratory diseases and gastroenteritis were used as a reference (4,5). Contacts with such symptoms would be tested for a range of pathogens to provide an alternative diagnosis. However, their removal from isolation would not be based solely on this testing because common pathogens are often detected in healthy controls.
Filovirus Evaluation in Contact Monitoring
Acute viremia develops in persons infected with Ebola virus, and viral antigens and RNA are detectable in serum, plasma, saliva, and occasionally other secreta (6,7). In early stages of infection, results of PCR-based assays have been positive 24–48 earlier than antigen-capture assays, making the PCR the method of choice. Although viral loads in severely ill patients are high, in the early course of illness, viral loads may be barely detectable (8). Therefore, proper evaluation of PCR-based methods, with particular emphasis on detection limits, is crucial for reliance on these diagnostics during monitoring. The filovirus diagnostics would therefore be conducted simultaneously in at least 2 laboratories. The Bernhard-Nocht-Institute for Tropical Medicine (BNI) in Hamburg, Germany, provided protocols for PCR-based detection of MARVs. They had been validated in a joint study between P4 laboratories, using all MARV isolates available in these laboratories as reference material (8).
Sequence analysis of the patient’s MARV strain showed it was most closely related to the first-identified Marburg virus isolate from Uganda, the Popp strain. Therefore, we assumed that detection limits reported for the Popp strain would apply to this strain as well. Reagent kits based on the Panning protocol were assembled at our request and kindly provided within a few days (Thomas Laue; QIAGEN, Hamburg, Germany). Evaluation of this kit, using extracts from patient serum and other possible sample types (throat swab, plasma, serum, feces), provided reliable results. Additionally, strain specific Taqman PCR was designed at the Department of Virology at the Erasmus University Hospital, with detection limits similar to those of the Panning protocol.
Laboratory Procedures Used in the Follow-up Survey
After inactivation and fixation on immunofluorescent antibody assay slides, the samples were stored at –20°C outside the high-containment laboratory, and further investigations using the inactivated virus were performed under BioSafety Level 2 conditions. Testing was performed using 1:10 and 1:40 dilutions in 1× phosphate-buffered saline of the contact sera, with positive (mouse monoclonal antibody against MARV) and negative (MARV-negative mouse sera) controls on every slide. In the initial screening, the presence of immunoglobulin (Ig) G and IgM was investigated by using IgM and anti-IgG secondary antibodies conjugated with fluorescein isothiocyanate (FITC).
After inconclusive results in the first screening, procedures were repeated using dilutions 1:20, 1:40, 1:80, 1:160, and 1:320 (plus negative and positive control) to enable identification of a potentially higher antibody titer. Inconclusive samples were double-stained with mouse monoclonal antibody and antibodies from the contact sera. The double fluorescence was detected by using 2 differently conjugated secondary antibodies: anti-mouse IgG-rhodamine and anti-human IgG-FITC to differentiate staining between virus particles.
In the initial evaluation of the slides, performed by 2 of the authors (P.E., S.D.), 2 samples could not clearly be identified as negative because they lacked the characteristic round virus inclusions in the cells. However, when virus particles in the infected cells were visualized using monoclonal antibody and overlaying it with the fluorescence of the human antibodies, all activity could be attributed to nonspecific background binding. No overlapping fluorescence of human antibody and mouse monoclonal antibody against Marburg hemorrhagic fever could be observed. Therefore, all tested sera were considered negative for IgG and IgM antibodies to MARV.
References
1. Jones SM, Feldmann H, Stroher U, Geisbert JB, Fernando L, Grolla A, et al. Live attenuated recombinant vaccine protects nonhuman primates against Ebola and Marburg viruses. Nat Med. 2005;11:786–90. PubMed DOI: 10.1038/nm1258
2. Daddario-DiCaprio KM, Geisbert TW, Stroher U, Geisbert JB, Grolla A, Fritz EA, et al. Postexposure protection against Marburg haemorrhagic fever with recombinant vesicular stomatitis virus vectors in non-human primates: an efficacy assessment. Lancet. 2006;367:1399–404. PubMed DOI: 10.1016/S0140-6736(06)68546-2
3. Geisbert TW, Daddario-DiCaprio KM, Williams KJ, Geisbert JB, Leung A, Feldmann F, et al. Recombinant vesicular stomatitis virus vector mediates postexposure protection against Sudan Ebola hemorrhagic fever in nonhuman primates. J Virol. 2008;82:5664–8. PubMed DOI: 10.1128/JVI.00456-08
4. Gageldonk-Lafeber AB, Heijnen ML, Bartelds AI, Peters MF, van der Plas SM, Wilbrink B. A case-control study of acute respiratory tract infection in general practice patients in the Netherlands. Clin Infect Dis. 2005;41:490–7. PubMed DOI: 10.1086/431982
5. de Wit MA, Koopmans MP, Kortbeek LM, Wannet WJ, Vinje J, van Leusden F, et al. Sensor, a population-based cohort study on gastroenteritis in the Netherlands: incidence and etiology. Am J Epidemiol. 2001;154:666–74. PubMed DOI: 10.1093/aje/154.7.666
6. Leroy EM, Baize S, Volchkov VE, Fisher-Hoch SP, Georges-Courbot MC, Lansoud-Soukate J, et al. Human asymptomatic Ebola infection and strong inflammatory response. Lancet. 2000;355:2210–5. PubMed DOI: 10.1016/S0140-6736(00)02405-3
7. Towner JS, Rollin PE, Bausch DG, Sanchez A, Crary SM, Vincent M, et al. Rapid diagnosis of Ebola hemorrhagic fever by reverse transcription-PCR in an outbreak setting and assessment of patient viral load as a predictor of outcome. J Virol. 2004;78:4330–41. PubMed DOI: 10.1128/JVI.78.8.4330-4341.2004
8. Panning M, Laue T, Olschlager S, Eickmann M, Becker S, Raith S, et al. Diagnostic reverse-transcription polymerase chain reaction kit for filoviruses based on the strain collections of all European biosafety level 4 laboratories. J Infect Dis. 2007;196(Suppl 2):S199–204. PubMed DOI: 10.1086/520600
AcknowledgmentsThis article was written on behalf of the members of the national response team, which included the authors and L. Isken, W. Ransz, and A. Jacobi, B. van der Walle and P. Willemse, R. Daemen and D. van Oudheusden, A. Brouwer and C. Bleeker, and T. Schmitt.
We acknowledge the invaluable support from our colleagues across the world, particularly H. Feldmann, R. Swanepoel, Matthias Niedrig, Thomas Laue, John Towner, E. Leroy, E. Gavrilin, R. Andraghetti, F. Plummer, T. Geisbert, G. Nabel, C.J. Peters, and B. Graham. We thank M. van der Lubben, H. Vennema, B. Wilbrink, G.J. Godeke, B. van der Veer, M. Timmer, B. Niemeijer, C. Burghoorn-Maas, T. Mes, G. van Willigen, E. Kuijper, M. Feltkamp, J. van Pelt, and M. Wulff for their assistance; Jim van Steenbergen for critical comments on the manuscript; and Lucy Phillips for editing.
Parts of this study were supported by a grant from the Dutch Research Foundation (ZonMw).
Biography• Dr Timen is a senior consultant in communicable disease control at the Centre for Infectious Diseases of the National Institute of Public Health and the Environment, the Netherlands. Her main research interest is the public health response to outbreaks and threats.
FootnotesSuggested citation for this article: Timen A, Koopmans MPG, Vossen ACTM, van Doornum GJJ, Günther S, van den Berkmortel F, et al. Response to imported case of Marburg hemorrhagic fever, the Netherlands. Emerg Infect Dis [serial on the Internet]. 2009 Aug [date cited]. Available
fromhttp://www.cdc.gov/EID/content/15/8/1171.htmReferences1. Crowcroft NS, Morgan D, Brown D Viral haemorrhagic fevers in Europe—effective control requires a coordinated response. Euro Surveill 2002;7:31–2 [PubMed]
2. Centers for Disease Control and Prevention Outbreak of Marburg virus hemorrhagic fever—Angola, October 1, 2004–March 29, 2005. MMWR Morb Mortal Wkly Rep2005;54:308–9 [PubMed]
3. Borio L, Inglesby T, Peters CJ, Schmaljohn AL, Hughes JM, Jahrling PB, et al.Hemorrhagic fever viruses as biological weapons: medical and public health management. JAMA 2002;287:2391–405 10.1001/jama.287.18.2391 [PubMed][Cross Ref]
4. Leffel EK, Reed DS Marburg and Ebola viruses as aerosol threats. Biosecur Bioterror2004;2:186–91 10.1089/bsp.2004.2.186 [PubMed] [Cross Ref]
5. Peters CJ Marburg and Ebola virus hemorrhagic fevers. In: Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. Philadelphia: Elsevier Inc.; 2005. p. 2057–9.
6. Siegert R Marburg virus. Virology monograph. New York: Springer-Verlag; 1972. p. 98–153.
7. Gear JS, Cassel GA, Gear AJ, Trappler B, Clausen L, Meyers AM, et al. Outbreak of Marburg virus disease in Johannesburg. BMJ 1975;4:489–93 10.1136/bmj.4.5995.489[PMC free article] [PubMed] [Cross Ref]
8. Johnson ED, Johnson BK, Silverstein D, Tukei P, Geisbert TW, Sanchez AN, et al.Characterization of a new Marburg virus isolated from a 1987 fatal case in Kenya. Arch Virol Suppl 1996;11:101–14 [PubMed]
9. Smith DH, Johnson BK, Isaacson M, Swanapoel R, Johnson KM, Killey M, et al.Marburg-virus disease in Kenya. Lancet 1982;1:816–20 10.1016/S0140-6736(82)91871-2 [PubMed] [Cross Ref]
10. Khan AS, Sanchez A, Pflieger AK Filoviral haemorrhagic fevers. Br Med Bull1998;54:675–92 [PubMed]
11. World Health Organization Marburg fever, Democratic Republic of the Congo. Wkly Epidemiol Rec 1999;74:145. [PubMed]
12. World Health Organization Viral haemorrhagic fever/Marburg, Democratic Republic of the Congo. Wkly Epidemiol Rec 1999;74:157–8 [PubMed]
13. World Health Organization Outbreak of Marburg haemorrhagic fever: Uganda, June–August 2007. Wkly Epidemiol Rec 2007;82:381–4 [PubMed]
14. European Network for Diagnostics of Imported Viral Diseases Management and control of viral hemorrhagic fevers [cited 2008 Aug 13]. Available
fromhttp://www.enivd.de15. Drosten C, Gottig S, Schilling S, Asper M, Panning M, Schmitz H, et al. Rapid detection and quantification of RNA of Ebola and Marburg viruses, Lassa virus, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, dengue virus, and yellow fever virus by real-time reverse transcription-PCR. J Clin Microbiol2002;40:2323–30 10.1128/JCM.40.7.2323-2330.2002 [PMC free article] [PubMed][Cross Ref]
16. Towner JS, Pourrut X, Albarino CG, Nkogue CN, Bird BH, Grard G, et al. Marburg virus infection detected in a common African bat. PLoS One 2007;2:e764 10.1371/journal.pone.0000764 [PMC free article] [PubMed] [Cross Ref]
17. Swanepoel R, Smit SB, Rollin PE, Formenty P, Leman PA, Kemp A, et al. Studies of reservoir hosts for Marburg virus. Emerg Infect Dis 2007;13:1847–51 [PMC free article][PubMed]
18. Filoviruses. In: Steenbergen JE, Timen A, Beaujean DJ, editors. LCI guidelines infectious disease control edition 2008 [in Dutch]. Bilthoven: National Institute for Public Health and the Environment; 2008. p. 478–84.
19. Haas WH, Breuer T, Pfaff G, Schmitz H, Kohler P, Asper M, et al. Imported Lassa fever in Germany: surveillance and management of contact persons. Clin Infect Dis2003;36:1254–8 10.1086/374853 [PubMed] [Cross Ref]
20. Borchert M, Muyembe-Tamfum JJ, Colebunders R, Libande M, Sabue M, van der Stuyft P Short communication: a cluster of Marburg virus disease involving an infant.Trop Med Int Health 2002;7:902–6 10.1046/j.1365-3156.2002.00945.x [PubMed][Cross Ref]
21. Borchert M, Mulangu S, Swanepoel R, Libande ML, Tshomba A, Kulidri A, et al.Serosurvey on household contacts of Marburg hemorrhagic fever patients. Emerg Infect Dis 2006;12:433–9 [PMC free article] [PubMed]
22. Bausch DG, Sprecher AG, Jeffs B, Boumandouki P Treatment of Marburg and Ebola hemorrhagic fevers: a strategy for testing new drugs and vaccines under outbreak conditions. Antiviral Res 2008;78:150–61 10.1016/j.antiviral.2008.01.152 [PubMed][Cross Ref]
23. Conrad JL, Isaacson M, Smith EB, Wulff H, Crees M, Geldenhuys P, et al.Epidemiologic investigation of Marburg virus disease, southern Africa, 1975. Am J Trop Med Hyg 1978;27:1210–5 [PubMed]
24. Bausch DG, Borchert M, Grein T, Roth C, Swanepoel R, Libande ML, et al. Risk factors for Marburg hemorrhagic fever, Democratic Republic of the Congo. Emerg Infect Dis 2003;9:1531–7 [PMC free article] [PubMed]
25. Centers for Disease Control and Prevention Interim guidance for managing patients with suspected viral hemorrhagic fever in US hospitals [cited 2008 Oct 13]. Available from
http://www.cdc.gov/ncidod/dhqp/bp_vhf_i ... dance.html26. Health Protection Agency Management and control of VHF (ACDP 1996) [cited 2008 Oct 13]. Available
fromhttp://www.hpa.org.uk/web/HPAwebFil ... 494738200527. Wirtz A, Niedrig M, Fock R Management of patients in Germany with suspected viral haemorrhagic fever and other potentially lethal contagious infections. Euro Surveill2002;7:36–42 [PubMed]