by Susanna C. Larsson, Edward Giovannucci, Alicja Wolk
July 13, 2006
© 2006 Wiley-Liss, Inc.
NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHT
YOU ARE REQUIRED TO READ THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
Abstract
Few prospective studies have examined the relationship between coffee consumption and risk of stomach cancer, and the findings have been inconsistent. We prospectively investigated the association of long-term coffee consumption with risk of stomach cancer in a population-based cohort study of 61,433 Swedish women. Information on coffee consumption was collected with a food-frequency questionnaire at baseline (1987–1990) and updated in 1997. During a mean follow-up of 15.7 years from 1987 through June 2005, 160 incident cases of stomach cancer were diagnosed. Coffee consumption was positively associated with the risk of stomach cancer. Compared to women who consumed 1 or fewer cups of coffee per day, the multivariate hazard ratios were 1.49 (95% = 0.97–2.27) for women who drank 2–3 cups per day and 1.86 (95% CI = 1.07–3.25) for those who drank 4 or more cups per day (p for trend = 0.01). An increase of 1 cup of coffee per day was associated with a statistically significant 22% increased risk of stomach cancer (hazard ratio = 1.22; 95% CI = 1.05–1.42). These prospective data suggest that coffee consumption may increase the risk of stomach cancer in a dose–response manner. This finding needs to be confirmed in other prospective studies.
Coffee contains numerous substances with potentially genotoxic and mutagenic properties,1, 2 which has raised the concern that high consumption of this beverage might increase the risk of various malignancies. The relationship of coffee consumption with the risk of stomach cancer has been the subject of several case-control studies, with largely null results,3 and a limited number of prospective cohort studies.4, 5, 6, 7 The findings from cohort studies have been mixed, with both positive4, 6 and inverse associations reported,7 but the association was statistically significant in only 1 study.6 The few existing cohort studies were based on a single assessment of coffee consumption at baseline and did not consider possible changes in diet during follow-up, which could lead to misclassification of long-term coffee consumption.
In Sweden, the average coffee intake per capita is one of the highest in the world.8 In this investigation, we analyzed prospective data from a large population-based cohort of Swedish women to examine whether long-term coffee consumption (based on repeated assessments) is associated with the risk of stomach cancer.
Material and Methods
Study population
The current investigation is based on data from the Swedish Mammography Cohort (SMC), a prospective population-based cohort study established between 1987 and 1990.9 The source population consisted of all women who were born between 1914 and 1948 and were residents of Västmanland and Uppsala counties of central Sweden from 1987 to 1990. A total of 66,651 women, representing 74% of the source population, returned a completed questionnaire about diet, weight, height and education. In the autumn of 1997, a second questionnaire was mailed to all 56,030 participants who were still alive and living in the study area, to update information on diet and to obtain data on other lifestyle factors; 39,227 women (70%) responded to this questionnaire. The study was approved by the Regional Ethics Committee at the Karolinska Institutet (Stockholm, Sweden).
We excluded at baseline participants who had implausibly high or low total energy intake (i.e., 3 standard deviations from the mean value for log-transformed energy intake), those with incorrect or missing national registration number and those with a cancer diagnosis other than nonmelanoma skin cancer before baseline. This left 61,433 women for these analyses.
Assessment of coffee consumption
Coffee consumption was assessed at baseline and in 1997 with a food-frequency questionnaire (FFQ). In these FFQs, participants were asked how often, on average, over the past year they had consumed coffee as well as other foods and beverages. The baseline FFQ had 8 predefined response categories, ranging from “never/seldom” to “4 or more times per day.” In the second FFQ, coffee consumption was inquired about as an open question. We converted the responses for coffee consumption to an average daily intake for each participant. To assess the validity of self-reported coffee consumption, estimated intake from the baseline FFQ was compared with those from four 1-week weighed diet records in a random sample of 129 women from the study population. The Spearman correlation coefficient on coffee consumption, as assessed by the FFQ compared with the diet records, was 0.63 (A. Wolk, unpublished data).
Assessment of nondietary exposures
The baseline questionnaire collected information on education, weight and height. Information on smoking status and history was obtained in the second questionnaire from 1997. Pack-years were estimated from smoking history by multiplying the average number of packs of cigarettes smoked per day by the number of years of smoking.
Case ascertainment and follow-up
We identified incident cases of stomach cancer (International Classification of Diseases, Ninth Revision, code 151) by computerized record linkage of the study cohort with the National and Regional Swedish Cancer registers, both of which provides almost 100% case coverage.10, 11 The Swedish Cancer Registry does not use information on cancers based on death certificates. Information on dates of death and dates of migration was obtained from the Swedish Death and Population registers at Statistics Sweden.
Statistical analysis
For each woman, person-time of follow-up was counted from the date of entry into the cohort to the date of diagnosis of stomach cancer, death, migration, or June 30, 2005, whichever came first. Participants were grouped into 3 categories according to their coffee consumption: ≤1, 2–3 and ≥4 cups/day (2–3 times per day was a predefined frequency category on the baseline FFQ and could not be split into 2 categories). Hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated using Cox proportional hazards models.12 To control for confounding by age and calendar time, we stratified the analysis jointly by age in months at baseline and by the year of entry into the cohort. All multivariate models were further adjusted for education (less than high school, high school graduate or more than high school), alcohol intake (0, 0.1 to <15, 15 to <30 or ≥30 g/week) and tea consumption (never/seldom, <1 cup/day or ≥1 cup/day). Body mass index (the weight in kilograms divided by the height in square meters) and intakes of total energy, processed meat, fruit and vegetables were excluded from the multivariate models, because they were not confounders in these analyses. In a subanalysis using information from the second questionnaire, we additionally controlled for smoking (never smoker, past smoker and <20 pack-years, past smoker and ≥20 pack-years, current smoker and <20 pack-years or current smoker and ≥20 pack-years). We tested the proportional hazards assumption for coffee consumption in relation to stomach cancer risk using the likelihood ratio test; the proportional hazards assumption was satisfied.
To provide a better estimate of the effect of long-term average coffee consumption and to reduce random within-person variation, we used a cumulative average method.13 Specifically, coffee consumption at baseline was used to predict stomach cancers diagnosed from baseline through 1997, and the average of the baseline and 1997 coffee consumption was used to predict outcomes from 1998 through June 2005. In a secondary analysis, baseline coffee consumption was used to predict stomach cancers diagnosed during the entire follow-up.
Tests for trend were conducted by modeling coffee consumption as a continuous variable. In addition, the continuous measure of cumulative updated coffee consumption was used to fit a restricted cubic spline model with 4 knots, and to obtain a smooth representation of the HR as a function of coffee consumption.14, 15 All analyses were performed with SAS software, release 9.1 (SAS Institute, Cary, NC). All reported p-values are 2-sided.
Results
Baseline characteristics of the study population according to coffee consumption are shown in Table I. At baseline, 18% of participants reported consuming 4 cups of coffee per day or more; 3.7% of participants reported that they never/seldom consumed coffee. Compared with women who consumed 1 or fewer cups of coffee per day (22.5%), those who drank 4 or more cups per day were younger and less likely to have a postsecondary education, but were more likely to smoke. In addition, women with greater coffee consumption drank less tea.
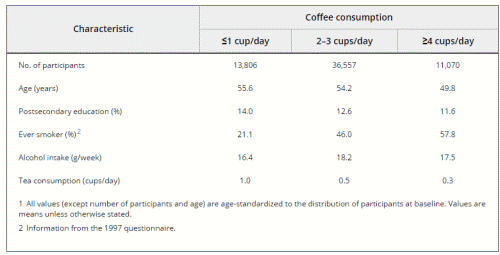
Table I. Baseline Characteristics of 61,433 Women in the SMC According to Coffee Consumption (1987–1990)1
Over a mean follow-up of 15.7 years (966,807 person-years), from 1987 through June 30, 2005, 160 incident cases of stomach cancer were diagnosed among the 61,433 participants. We found a statistically significant positive association between coffee consumption and stomach cancer risk (Table II). After adjusting for age, education, alcohol and tea consumption, women who drank 4 or more cups of coffee per day had a statistically significant 86% increased risk of stomach cancer when compared with those who drank 1 or fewer cups of coffee per day (for cumulative updated coffee consumption, HR = 1.86; 95% CI = 1.07–3.25; p for trend = 0.01). Excluding the first 3 years of follow-up did not alter the results appreciably (HR = 1.77; 95% CI = 0.97–3.21). When we split the lowest category of coffee consumption into 2 categories (<1 and 1 cup/day) and used the lowest category (<1 cup/day) as reference group, the multivariate HRs for increasing categories of coffee consumption (1 cup/day, 2–3, cups/day, and ≥4 cups/day) were 1.17 (95% CI = 0.51–2.66), 1.66 (95% CI = 0.80–3.44) and 2.07 (95% CI = 0.92–4.67).
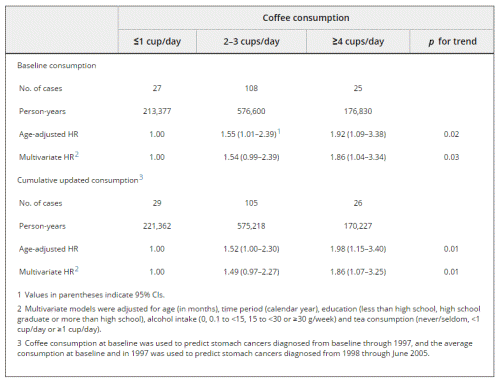
Table II. Hazard Ratios of Stomach Cancer According to Baseline and Cumulative Updated Coffee Consumption Among 61,433 Women in the SMC (1987–2005)
The restricted cubic spline curve indicated that the relation between coffee consumption (cumulative updated) and stomach cancer risk was linear (Fig. 1). In a multivariate analysis, an increment of 1 cup of coffee per day was associated with a 22% increased risk of stomach cancer (HR = 1.22; 95% CI = 1.05–1.42).
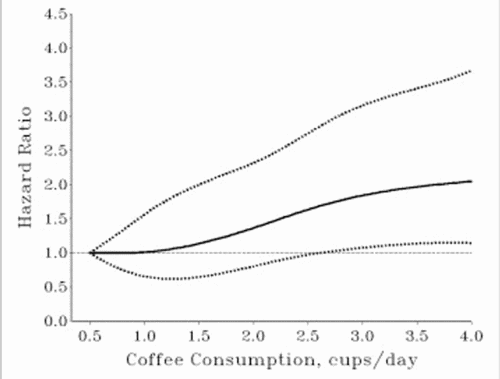
Figure 1. Multivariate HRs of stomach cancer according to cumulative updated coffee consumption among 61,433 women in the SMC (1987–2005). Data were fitted by a restricted cubic spline Cox proportional hazards model. HRs were adjusted for age (in months), time period (calendar year), education (less than high school, high school graduate or more than high school), alcohol intake (0, 0.1 to <15, 15 to <30 or ≥30 g/week) and tea consumption (never/seldom, <1 cup/day or ≥1 cup/day). The dotted lines represent 95% CIs.
In a subanalysis using data from the second questionnaire, we examined whether further control for smoking had any effect on our results. In this analysis (including 55 incident cases), the HRs of stomach cancer for a daily increment of 1 cup of coffee were 1.23 (95% CI = 0.98–1.56) after adjusting for age, education, alcohol intake and tea consumption, and 1.21 (95% CI, 0.96–1.54) after additional adjustment for smoking.
Discussion
In this large population-based prospective study of Swedish women, coffee consumption was significantly associated with risk of stomach cancer in a dose–response manner. We observed an 86% increase in stomach cancer risk for those who drank 4 or more cups of coffee per day.
Our findings are consistent with a previous prospective cohort of 11,906 Japanese residents of Hawaii (with 108 incident cases), which showed that compared with nondrinkers, those who consumed 2 or more cups of coffee per day had a statistically significant 1.8-fold higher risk of stomach cancer.6 In a cohort of 16,555 Norwegian women and men (with 147 stomach cancer deaths), a 1.5-fold increased risk of stomach cancer was observed for the highest (≥7 cups/day) compared with the lowest category (≤2 cups/day) of coffee consumption, but the findings were not statistically significant.4 In contrast, another Norwegian cohort study of 21,238 women (32 incident cases) and 21,735 men (46 incident cases) showed a nonsignificant inverse association between coffee consumption and stomach cancer risk.7 Coffee consumption was not related to stomach cancer risk in a cohort of 7,355 Japanese women and men (106 incident cases).5 Case-control studies have not reported any significant relationship between coffee consumption and risk of stomach cancer.3 Results from case-control studies, however, have to be evaluated with caution because of the retrospective assessment of diet. Although cases are asked about their diet from the time before their cancer diagnosis, their recall of coffee consumption may be affected by their current intake and by their disease status.
The biological mechanism behind the observed positive association of coffee consumption with stomach cancer risk is not clear. Coffee is a complex chemical mixture that contains many substances that might be mutagenic or carcinogenic.1, 2 For example, coffee contains caffeic acid and chlorogenic acid, which have been shown to be strong inhibitors of DNA methylation in vitro.16 Aberrant DNA methylation is an early event in stomach carcinogenesis.17, 18 Coffee consumption has been associated with an increased risk of chronic atrophic gastritis,19 which is considered to be one of the most important pre-cancerous states of the stomach.20 Both regular and decaffeinated coffee have been demonstrated to stimulate release of the gastrointestinal peptide hormone gastrin.21 There is evidence that gastrin may be implicated in the development of stomach cancer.22, 23 Significantly higher plasma gastrin levels have been found in stomach cancer patients compared with healthy controls.24, 25 It has been suggested that elevated gastrin levels may be not only a consequence but also a cause of gastric atrophy.22
This study has several major strengths. First, the prospective design avoided the potential for differential recall of coffee consumption by stomach cancer cases and noncases, because exposure information was collected before any cancer was diagnosed. Second, the practically complete follow-up of our study population through computerized linkage to various Swedish population-based registers minimizes the concern that our findings are affected by differential loss to follow-up. Third, we had repeated measures of diet and were able to update information on coffee consumption during follow-up to obtain a better estimate of the effect of long-term average coffee consumption and reduce random within-person variation.
A potential limitation to this study is an inability to examine whether the association with coffee consumption varies according to anatomical subsite or histological subtype, or whether the association is modified by Helicobacter pylori infection status. Another limitation is that information on smoking was not collected at baseline. However, in a subanalysis using data from the second questionnaire, the positive relation between coffee consumption and stomach cancer persisted after adjusting for smoking. As in any observational study, measurement error is of concern because we relied on self-reported information on coffee consumption. Such error is most likely to have been nondifferential because of our prospective design and may have resulted in attenuation of risk estimates.
In our study, we did not have information on the type of coffee, i.e., whether it was filtered or boiled, caffeinated or decaffeinated. A previous study26 indicated that in Sweden (in Stockholm and Västernorrland counties), among women who drank coffee, about 85% consumed filtered coffee, 8.5% boiled coffee, and 6.5% consumed both filtered and boiled coffee. Decaffeinated coffee is very uncommon in Sweden.
In summary, findings from this population-based cohort of Swedish women suggest that coffee consumption is associated with an increased risk of stomach cancer in a dose–response manner. However, our observational study cannot prove a cause–effect relationship, and it is premature to recommend decreased coffee drinking as a means to reduce the risk of stomach cancer. Further prospective studies are needed to confirm this relationship.
_______________
References
1. Ames BN, Gold LS. Environmental pollution, pesticides, and the prevention of cancer: misconceptions. FASEB J 1997; 11: 1041–52.
2. Nehlig A, Debry G. Potential genotoxic, mutagenic and antimutagenic effects of coffee: a review. Mutat Res 1994; 317: 145–62.
3. World Cancer Research Fund and American Institute for Cancer Research. Food, nutrition and the prevention of cancer: a global perspective. Washington, DC: American Institute for Cancer Research, 1997.
4. Jacobsen BK, Bjelke E, Kvale G, Heuch I. Coffee drinking, mortality, and cancer incidence: results from a Norwegian prospective study. J Natl Cancer Inst 1986; 76: 823–31.
5. Nomura A, Heilbrun LK, Stemmermann GN. Prospective study of coffee consumption and the risk of cancer. J Natl Cancer Inst 1986; 76: 587–90.
6. Galanis DJ, Kolonel LN, Lee J, Nomura A. Intakes of selected foods and beverages and the incidence of gastric cancer among the Japanese residents of Hawaii: a prospective study. Int J Epidemiol 1998; 27: 173–80.
7. Stensvold I, Jacobsen BK. Coffee and cancer: a prospective study of 43,000 Norwegian men and women. Cancer Causes Control 1994; 5: 401–8.
8. Fredholm BB, Battig K, Holmen J, Nehlig A, Zvartau EE. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol Rev 1999; 51: 83–133.
9. Wolk A, Bergström R, Hunter D, Willett W, Ljung H, Holmberg L, Bergkvist L, Bruce A, Adami HO. A prospective study of association of monounsaturated fat and other types of fat with risk of breast cancer. Arch Intern Med 1998; 158: 41–5.
10. Ekström AM, Signorello LB, Hansson LE, Bergström R, Lindgren A, Nyrén O. Evaluating gastric cancer misclassification: a potential explanation for the rise in cardia cancer incidence. J Natl Cancer Inst 1999; 91: 786–90.
11. Mattsson B, Wallgren A. Completeness of the Swedish Cancer Register. Non-notified cancer cases recorded on death certificates in 1978. Acta Radiol Oncol 1984; 23: 305–13.
12. Cox DR, Oakes D. Analysis of survival data. London: Chapman & Hall, 1984.
13. Hu FB, Stampfer MJ, Rimm E, Ascherio A, Rosner BA, Spiegelman D, Willett WC. Dietary fat and coronary heart disease: a comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am J Epidemiol 1999; 149: 531–40.
14. Durrleman S, Simon R. Flexible regression models with cubic splines. Stat Med 1989; 8: 551–61.
15. Greenland S, Michels KB, Robins JM, Poole C, Willett WC. Presenting statistical uncertainty in trends and dose-response relations. Am J Epidemiol 1999; 149: 1077–86.
16. Lee WJ, Zhu BT. Inhibition of DNA methylation by caffeic acid and chlorogenic acid, two common catechol-containing coffee polyphenols. Carcinogenesis 2006; 27: 269–77.
17. Kang GH, Shim YH, Jung HY, Kim WH, Ro JY, Rhyu MG. CpG island methylation in premalignant stages of gastric carcinoma. Cancer Res 2001; 61: 2847–51.
18. Lee JH, Park SJ, Abraham SC, Seo JS, Nam JH, Choi C, Juhng SW, Rashid A, Hamilton SR, Wu TT. Frequent CpG island methylation in precursor lesions and early gastric adenocarcinomas. Oncogene 2004; 23: 4646–54.
19. The Eurohepygast Study Group. Risk factors for atrophic chronic gastritis in a European population: results of the Eurohepygast study. Gut 2002; 50: 779–85.
20. Correa P. Human gastric carcinogenesis: a multistep and multifactorial process––First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res 1992; 52: 6735–40.
21. Boekema PJ, Samsom M, van Berge Henegouwen GP, Smout AJ. Coffee and gastrointestinal function: facts and fiction. A review. Scand J Gastroenterol Suppl 1999; 230: 35–9.
22. Aly A, Shulkes A, Baldwin GS. Gastrins, cholecystokinins and gastrointestinal cancer. Biochim Biophys Acta 2004; 1704: 1–10.
23. Evers BM. Gastrointestinal growth factors and neoplasia. Am J Surg 2005; 190: 279–84.
24. Rakic S, Hinder RA, Adanja G, DeMeester TR. Elevated serum gastrin levels in patients with gastric cancer. J Surg Oncol 1991; 47: 79–81.
25. Triantafillidis JK, Merikas E, Govosdis V, Konstandellou E, Cheracakis P, Barbatzas C, Tzourmakliotis D, Peros G. Increased fasting serum levels of growth hormone and gastrin in patients with gastric and large bowel cancer. Hepatogastroenterology 2003; 50 ( Suppl 2): cclvi–cclx.
26. Hammar N, Andersson T, Alfredsson L, Reuterwall C, Nilsson T, Hallqvist J, Knutsson A, Ahlbom A. Association of boiled and filtered coffee with incidence of first nonfatal myocardial infarction: the SHEEP and the VHEEP study. J Intern Med 2003; 253: 653–9.
