The Good, the Bad and the Ugly: a review of SARS Lab Escapes
by Gilles Demaneuf
Nov 16, 2020
Research by DRASTIC, a group of researchers working together to investigate the origins of SARS-CoV-2, starting from facts and not bending to any pressure. DRASTIC members were the first ones to discover the Mojiang ‘miners’ story.
DOI: 10.5281/zenodo.4293257
Introduction
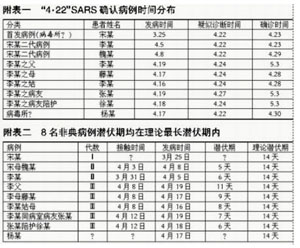
In 2003–04, in the wake of the SARS epidemics, there were multiple cases of laboratory acquired infection (LAI) with SARS in just a few months: first in a P3 in Singapore, then in a P3 lab that was part of a military P4 complex in Taipei, and last a protracted case in a P3 in Beijing. The ‘WHO SARS Risk Assessment and Preparedness Framework’ has a good summary of these lab accidents:
Since July 2003, there have been four occasions when SARS has reappeared. Three of these incidents [note: Singapore, Taipei and Beijing] were attributed to breaches in laboratory biosafety and resulted in one or more cases of SARS. The most recent laboratory incident [note: in Beijing] resulted in 9 cases, 7 of which were associated with one chain of transmission and with hospital spread. Two additional cases at the same laboratory with a history of illness compatible with SARS in February 2004 were detected as part of a survey of contacts at the facility. [i.1]
This article reviews some of these cases and discusses briefly some of the insights that were gained from these at the time.
1. The Good: Singapore P3 — Sep 2003
In a few words: This first SARS LAI accident (in Singapore) exposed some serious structural biosafety issues but the investigation was very thorough and transparent, and even extended into a review of all BSL-3s in the city-state. The Singapore government used it as an opportunity to fundamentally redesign its biosafety approach, just at the time when more BSL-3s were due to come in line. We also note that the (strongly controlled) local media do not seem to have contributed any additional details to the story.
1.1 The Accident:
In September 2003, a 27-year-old student from the National University of Singapore (NUS) was found to be infected with the SARS virus due to improper experimental procedures. [s.1]
The student was in his third year of a doctoral program in microbiology and was studying the replication of flaviviruses such as the West Nile Virus (which was just causing a peak of infection in the US at the time) in cells using electron microscopy. So far he had worked at the NUS BSL- 2 with the attenuated Sarafend strain of the West Nile virus. Because he wanted to compare this attenuated train with the more pathogenic New York strain of the virus (that one a BSL-3 pathogen), and since NUS had no BSL-3, an agreement was made for him to do this work at the BSL-3 facility of Institute of the Environmental Health Institute (EHI) of Singapore.
The BSL-3 of the EHI had been heavily involved in SARS research during the recent outbreak and was continuing that work at the time [s.6].
The student had no previous experience working in a BSL-3. On his first visit to the EHI, he was given only 20 minutes training with BSL-3 procedures before entering the BSL-3 lab.
On his second and last visit to the institute (Saturday 23 Aug 2003) he entered the lab 3 times:
• The first time, he went in with the technician, wearing only street clothes, and did not engage in any work.
• The second time, staff of the EHI were meeting. Before going to that staff meeting, the virology technician had grown up a stock of the New York isolate and centrifuged the supernatant from infected cells. The technician had then placed the centrifuge tubes in the Class II biological safety cabinet for the student to ampoule as a seed stock for his research. Accordingly the student put on a gown and two sets of gloves before entering the laboratory alone, where he spent 20 minutes unsupervised, transferring the cell supernatant into pre-labelled cryovials under the Class II biosafety hood. [s.6]
• The third time he re-entered the lab with the technician who was back from her meeting and transferred the cryovials to a –70°C freezer located in the BSL-2 facility, as no such freezer existed inside the BSL-3 laboratory, so that all frozen BSL-3 virus strains were effectively stored in the BSL-2 freezer.
The student did not engage in any more work with these before falling ill 3 days later on Aug 26, developing
The student did not engage in any more work with these before falling ill 3 days later on Aug 26, developing mild SARS symptoms. He sought outpatient medical care from his general practitioner, Singapore General Hospital (SGH) emergency room and a Chinese physician. He eventually returned to the SGH with persistent fevers and was admitted to the hospital on Sep 3. Fortunately he recovered and there were no secondary cases.
1.2 The Investigation:
Analysis of the ampoules of seed West Nile virus that the student stored on that 23rd Aug showed that the vials contained SARS- CoV as well as West Nile virus. That SARS-CoV matched the SARS isolate that was handled at the EHI.
Poor record keeping made it difficult to ascertain if there was a live SARS virus in the BSL-3 laboratory on that exact 23rd Aug, but it was established that there was some there 2 days before. [s.8]
In other words there had been a SARS-contamination within the BSL-3.
A 11-member review panel led by Antony Della-Porta, Biosafety Expert for the WHO [t.3], produced a report for the Ministry of Health in Singapore. The panel concluded that a combination of
“inappropriate laboratory standards and a cross-contamination of West Nile virus samples with SARS coronavirus in the laboratory led to the infection of the doctoral student”.[s.8]
The investigators also documented a variety of shortcomings within the EHI BSL-3 lab that most likely contributed to the incident. These included inadequate record-keeping procedures, totally inadequate training, inexistent virus stock inventory, patchy maintenance records plus a variety of structural problems including the absence of gauges to indicate the pressure differentials, the lack of a freezer to store samples, problems with HEPA filters and air supply, and other equipment deficiencies. [s.5, s.8].
Accordingly they first recommended that all BSL-3 work cease there ‘until the laboratory deficiencies have been addressed and subjected to external audit’. [s.8]
Note: While many circumstantial evidences pointed to a real risk of SARS contamination of the EHI BSL-3 lab, the exact causes of this particular accident — such as a precise bad manipulation or the failure of a precise piece of equipment — was never precisely identified.
1.3 Structural Issues:
At the invitation of the Singapore Ministry of Health, the investigation team went further than the EHI P3 lab and looked also at overall biosafety practices in other Singapore institutions. This was both a very courageous and very useful initiative, especially in view of the large number of new Singapore BSL-3 labs that were supposed to come in line soon at the time.
The Singapore General Hospital BSL-3, the NUS BSL-2 and the Defence Science Organisation were reviewed. The panel found deficiencies at the Singapore General Hospital as well as at the NUS BSL-2. [s.8] and made recommendations to improve biosecurity in each institution they visited.
More generally, the panel concluded that there were no appropriate standards, regulations or guidelines on biosafety in Singapore, and offered some recommendations [s.8] as to what could be done to rectify the situation:
Biological Standards
Recommendation 7.1: Need for a National Legislative basis for Standards in Biosafety Laboratories for Singapore.[..].
Recommendation 7.2: A structure should be created for laboratory certification covering both structure integrity and operating procedures. [..]
Recommendation 7.3: Creation of the tracking system for importation, exportation to and from Singapore. [..]
1.4 The Aftermath:
At the end of September, Lim Ruisheng, Minister of Environment of Singapore, apologised to the people of Singapore for the SARS infection incident in the laboratory of the Institute of Environmental Health. Lin Ruisheng said:
“The Environmental Health Research Institute must take responsibility, and the National Environmental Administration must also take responsibility. As the Minister of Environment, I should be more responsible. Because the investigation results of the investigation team show that our laboratory is indeed not safe enough.” [s.3]
Since 2003, Singapore has introduced legislation and regulations to cover the handling of high risk infectious agents and developed a laboratory accreditation system. Aligning incentives with regulation and training, they have also taken a number of initiatives to reward outstanding safety practices.[s.6]
2. The Bad: Taiwan Military P4 — December 2003
Ina few words: The second SARS LAI accident (Taiwan) was a less straightforward affair. First the human factor played a big role in delaying notification to the authorities. Then the accident had a large epidemic potential as it involved international air-travelling shortly after infection. Also, while the official reporting of the accident was generally transparent, a few aspects of the story remained somewhat opaque due to the military setting.
Taiwanese media, and interestingly also mainland China media, have contributed circumstantial details to the story. Generally, the reaction of the authority seems to have been earnest and constructive.
2. 1 The Accident:
Oddly most references to this lab accident simply fail to mention that it happened in a military P4 laboratory (likely built or equipped by France) utilising type III glove ported isolators [t.17]. These facts are particularly important and should be better publicised.
This lab escape involved the Taiwan Military Institute of Preventive Medical Research (IPMR) of the National Defence University. It is located in Baiji Mountain, Sanxia (or Sanhsia, 三峽, Three Georges), on the outskirts of Taipei Town, ands is allegedly partly dug into the hill. The laboratory is known as the centre of Taiwan’s biochemical capacities. The facility was allegedly capable of producing weapons-grade biological agents [t.1] and may have done so in the past [t.18], although Taiwanese officials claim it exists only to research ways of preventing biological attacks from mainland China.
According to unverified Chinese sources, Taiwan purchased the IPMR from France in 1983 [t.2]. A more recent Western source described it as being ‘full of advanced French-made equipment’ [t.18]

IPMR entrance
As the top biosafety lab in Taiwan, the IPMR has been racing against the clock to analyze the characteristics of the virus since the SARS outbreak. In May 2003 President Chen Shui-Bian had announced that he had ordered the IPMR to work on a vaccine for SARS. [t.18, t.20]
On December 6th 2003, 44-year-old Lieutenant-Colonel Chan Jiacong , a Ph.D. in pathology from Johns Hopkins University (US), contracted SARS at the IPMR due to negligence.
The researcher worked with SARS. He was cleaning his Taipei lab when he found a ripped bag [t.7] in the negative-pressure transport cabinet of the laboratory. He assumed that the bag had been leaking for several days and may have thought the virus had already lost its effectiveness. In any case the researcher was working alone and had to travel to Singapore the next day to address a meeting about his SARS research, so he was in a rush t.4, t.10]. [Note: we were unable to identify a suitable conference in Singapore on those days — suggestions welcome]
The proper way of cleaning that spill was to use vaporised hydrogen peroxide, but this would have taken hours. Instead he decided to use a shortcut in the form of 70% ethanol which he could spray on the spill before wiping it out [t.17].
As he could not reach the spot with the gloves attached to the cabinet, he instead wore a “normal mask” and surgical gloves, no protective gown, opened the transport cabinet, tried to reach the spill — but because he still could not reach it, he then put his head inside the cabinet pass-through to spray the spill with the ethanol mix. After spraying it and waiting for 10 minutes, he wiped the spill out.
In the process it seems that he then simply dumped the leaking bag on his trash cart.[t.2, t.7, t.8, t17].
“He was in a hurry to get ready for a conference in Singapore, so he was rushing to finish his disinfection work and was careless”
-- Su Ih-jen, chief of Taiwan’s Center for Disease Control [t.5]
Another source notes that:
[The] laboratory was also found to have violated many safety and record-keeping standards. For example, this researcher regularly worked long shifts (12 to 14 hours) usually alone and there was no timely procedure in place for reporting incidents. In addition, there was no record of him actually working in the laboratory since he had recently lost his building access card and was using a borrowed card on the date of the incident.
Della-Porta explains that the recommended procedure for decontamination of the safety cabinets used in the Taiwan facility is the use of a hydrogen peroxide generator, which takes several hours. He adds that a shortcut of 70 percent ethanol for 10 minutes is completely inadequate. In addition, there were inadequate standard operating procedures and Taiwan was without guidelines or regulations related to biological safety.’ [t.10]
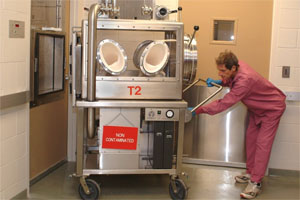
Example of a mobile Class III BSC (Biosafety Cabinet) — suitable for a BSL-4
On the following day, Dec. 7, he flew to Singapore with six laboratory colleagues on a China Airlines flight. He complained of fatigue and muscle pain on Dec. 8 while in Singapore but had no fever until the night of Dec. 10 after he returned to Taipei. He later had diarrhoea.
Soon after returning from Singapore, he reported to the Defence University clinic that he had developed influenza. Among many issues, the WHO investigation team recorded a failure to have a medical monitoring programme in place. Indeed senior management could not explain why, after Chan had reported to the Defence University clinic with a respiratory disease, he was not followed up for the next 6 days when he was absent from work. [t.16]
2.2 Fear of Losing face and bringing shame:
Faced with the standard symptoms of SARS, Lieutenant-Colonel Chan was very aware that he may have caught SARS. However he decided to self-isolate at home, relying on his father to care for him, as he was unwilling to seek medical care because he dreaded bringing disgrace to himself and his institution.
His father eventually persuaded him [t.6] to seek medical attention by threatening to commit suicide. He checked into Taiwan Hoping Hospital only on Dec 16 by which time he had developed other SARS symptoms, such as a cough and signs of pneumonia. He was readily diagnosed as having SARS [t.16].
His father said in a television interview that his son knew early on that he might have SARS. But he delayed going to the hospital because he didn’t want to panic the public and disgrace Taiwan. He quarantined himself at home until Dec. 16 and relied on his father for food and other care.
His father told CTI cable TV that his son: “wanted to die at home because he feared his illness would bring shame to his lab and the country”.
The father said he pleaded with his son to seek treatment. As the scientist’s condition worsened, his father said he threatened to commit suicide if he didn’t get help. “He finally agreed to go to the hospital after I threatened to kill myself,” his father said. “He was thinking of the country. He’s a military man, and military men are bound by a natural duty.” [t.6]
2.3 Reaction from the Authorities
A chest x-ray showed pneumonia in his right lung and polymerase chain reaction tests of throat and blood samples were positive for the SARS virus. The finding was further confirmed on multiple samples in two laboratories in Taipei.
Six colleagues were ordered to suspend work at the laboratory and start self-isolation at home for 21 days. The four family members of the officer self-isolated at home for 21 days, although they had not displayed fever or any other SARS-like symptoms.
Although Chan was not in the contagious phase when he travelled, around 95 people who had been in contact with him were quarantined until Dec. 23, including 19 that were on his flights and 79 in Singapore. None of them were infected. It is worth noting that officials were having difficulty contacting three Americans, a Japanese and a Singaporean who had been on the flight. Additionally two of his colleagues went to Singapore with the researcher on Dec. 7 and later traveled to the United States.[t.6, t.14]
Five foreigners — three Americans, one Japanese and a Singaporean — who sat near him on the return flight from Singapore have been located and told to monitor their health, officials said on Friday. The Japanese traveler was located in Taiwan where he lives, two of the Americans went to Hong Kong and the other is in the United States, said Shih Wen-yi, a spokesman for Taiwan’s Center for Disease Control. They were told to be on alert for SARS symptoms, like a fever. [t.6]

Lieutenant Colonel Chan leaving hospital
The Taiwan Science Council closed the IPMR temporarily and issued a resolution stating that Lieutenant Colonel Chan violated the “Laboratory Safety Guidelines and Norms” of the SARS project research plan and was given a sanction not to apply for research project funding.
The laboratory conducting severe acute respiratory syndrome (SARS) research at the Institute of Preventive Medicine Research (IPMR) of the National Defence Medical College was closed down temporarily Wednesday after one of its researchers was confirmed to have contracted the flu-like disease, military sources said.[t.6]
“All SARS research work in the military has also been halted”
-- Chang Sheng-yuan, director-general of Taiwan’s Military Medicine Bureau
The CDC conducted two complete environmental disinfections. All equipment was inspected in detail, and all personnel were retrained. [t.2, t.13]
Separately in China, authorities ordered all researchers to hand in SARS samples as a precaution. China’s Health Ministry told laboratories to send samples to “designated places for storage” and “demanded that all regions strengthen management of the SARS virus,” the official Xinhua News Agency said. [t.6]
2.4 WHO enquiry and international cooperation:
A WHO Team was invited to investigate the case. Anthony Della-Porta, who had earlier headed the WHO investigation of the SARS LAI assisted with the investigation.
As far as we can tell, the WHO investigation seems to have been rather comprehensive and transparent, finding and reporting quite a few failures, with the exact circumstances of the accident well delineated [t.13]. The international cooperation effort that followed seems to have been constructive and certainly helped rebuild trust in the Taiwanese capacity to manage their BSL3/4s:
In January 2004, the World Health Organization (WHO) dispatched an expert team [of 3] led by Dr. Antony Della-Porta to Taiwan because of the SARS case (Figure 1). They visited five existing and new constructions of BSL-3 laboratories during their short stay. In addition to the advice given to those individual laboratories, four major measures in terms of general management of biological safety were suggested.
Soon after, CDC invited two world-class experts in the field, Dr. Thomas Ksiazek from U.S. CDC and Dr. Kazuyoshi Sugiyama from National Institute of Infectious Diseases (NIID) of Japan, respectively, in March and November 2004, to give [Taiwan CDC] us a helping hand in inspecting current safety and management of equipment in laboratories of Biosafety Level 3 or above across Taiwan (Figure 2). [t.9]

Figure 1: WHO expertise committee investigated BSL-3 Virus laboratory of Taiwan CDC

Figure 2: Taiwan CDC invited experts from USA CDC and Japan NIID to Taiwan to assist in appraising BSL-3 laboratories
As the Taiwan CDC would later state,
‘The laboratory-acquired case of SARS in Taiwan sent a shockwave through the nation. However, this incident was a crucial turning point. It led to the seeking and adoption of advice from both national and international experts and scholars and, therefore, acquired invaluable learning experience in biological safety management’. [t.11]
2.5 Lessons to be learnt:
Henk Bekedam, the WHO representative in China, told reporters in Beijing that the case in Taiwan should alert scientists:
“It’s a clear reminder again that we have to be extremely cautious working with the SARS coronavirus and there are whole issues about whoever in the whole world is keeping a SARS specimen has to be very careful in dealing with this.” [t.6]
Anthony Della-Porta was even more clear:
“It hasn’t been a disaster, but it could have been.” [t.12]
As noted by Furmanski [t.15] (quoted here), this second outbreak further shook the virology communities in Asia, where many labs held and worked on SARS samples. On December 18, 2003 WHO released a new protocol for handling SARS specimens in the post-outbreak period, with special emphasis on reducing risk of and performing surveillance to detect laboratory infections. Although this protocol was clearly created after the first (Singapore) escape, the WHO chose its words carefully so as to avoid offending members but resolutely highlighted the laboratory risk-factor in its introduction:
“The possibility that a SARS outbreak could occur following a laboratory accident is a risk of considerable importance, given the relatively large number of laboratories currently conducting research using the SARS-CoV or retaining specimens from SARS patients. These laboratories currently represent the greatest threat for renewed SARS-CoV transmission through accidental exposure associated with breaches in laboratory biosafety.”
-- WHO [t.16]
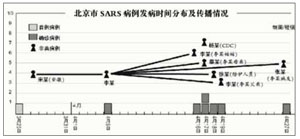
3. The Ugly: Beijing P3 — February and April 2004
Ina few words: The third lab incident is a very unsatisfactory affair. It happened in the context of a rather toxic ivory-tower academic system leading to unchallenged bad practices. Very limited official information was released when it ever was; effectively the Chinese government, and consequently the investigation report, provided very little insights or simply did not discuss key issues.
There are also some reasons to suspect an internal cover up of the first two SARS cases, and last (and not least), the sanctions announced by the Chinese Ministry of Health seem to have been more symbolic than anything else.
Without exaggeration, the Beijing LAI accidents and the subsequent outbreak seem very much to be a story of towering academic ego, shocking incompetence, obstruction of the truth and lack of accountability.
More importantly the unchallenged poor-handling of the investigation by the Chinese authorities — after the WHO left the scene — may have set a bad precedent.
Partially redeeming this dark picture, it is worth noting that the mainland China media contributed some very good pieces of investigative journalism which can still be found today online and deliver many essential clues as to what actually happened.
3.1 Setting the wrong expectations:
Following the Singapore SARS lab accident it became clear that laboratories handling SARS may not be as safe as one could expect, especially in nations rushing to build more P3 labs. This raised the question of the safety of the laboratories handling SARS in China.
Around the 13th Oct 2003 (so before the SARS accident in Taiwan) the National Health and Family Planning Commission of the People’s Republic published an article where it stated that:
“The nation’s SARS virus laboratories are safe. At present, no SARS patient has been infected from a research laboratory virus.” [b.4, b.13]
But the article went on to list reasons to be anxious and to make sure that the nation redoubles its effort to better control all risk factors:
According to Professor Zhu Qingyu, a P3 qualification only refers to the laboratory’s compliance with physical protection standards. In fact, the human factor comes first in terms of laboratory safety.
“Without good quality and sense of responsibility, (scientific research institutions) without strict and perfect management, being designated a P3 laboratory alone will not work.“ [b.13]
On December 18 2003, two days after the revelation of the Taiwan SARS lab accident, the WHO called for vigilance in P3 settings studying SARS and issued its ‘biosafety guidelines for handling of SARS-CoV specimens and cultures’ [t.16].
On the same day, the Chinese Ministry of Health duly followed suit and issued an emergency notice requesting all localities to strengthen the centralised management of infectious SARS virus strains and human specimens to ensure the biological safety of virus laboratories and storage units [b.12].
The Ministry of Science and Technology and the Ministry of Health also conducted safety inspections of the 15 to 20 P3 laboratories across the country at the end of December and started addressing the issue of labs that were handling SARS without approval. [b.1, b.12]
Incidentally, the CDC Institute of Virology in Beijing, being the most famous P3 lab handling SARS in the country, was explicitly mentioned in the press at the time:
P3 laboratories have corresponding strict operating procedures. [ — ]. The current research on SARS by the Institute of Virology of the China Center for Disease Control and Prevention is under such a strict operation. SARS laboratories implement a dual access system: first, the procedures must be complete; second, entering the laboratory must be approved by management personnel.
It is reported that in the laboratory of the Institute of Virology of the Chinese Center for Disease Control and Prevention, there are six or seven researchers engaged in SARS research, and these people can be directly exposed to the SARS virus. According to Ruan Li [director of the institute], in accordance to the rules, when researchers enter the laboratory, they must first pass through an air-lock, then through the anti-contamination area, and finally through another air-lock. The same is true when one comes out.
All viruses are wiped out or removed in the process, and they cannot escape the laboratory. Therefore, the key lies in whether the staff strictly follow the procedures. [b.12]

Article from the 19th Dec 2003
On the 19th Dec, Qi Guoming, director of the Science and Education Department of the Ministry of Health of China, detailed the actions taken by his ministry in an interview with a reporter from CCTV:
The Ministry of Health is now dealing with these problems. The laboratories that began to conduct SARS virus research without approval are being cleaned up and checked.
He explained that the review teams will examine the laboratory’s hardware, personnel quality, and the management of virus samples. Those laboratories that do not meet the standards will immediately stop research. [ — ]
‘If an accident occurs and infection is caused, it is a crime against the people and society’. [b.12]
3.2 Flagship role of the CDC Institute of Virology:
The National Institute for Viral Disease Control and Prevention (NIVDC) in Beijing (病毒病预防控制所), in short the Institute of Virology, is part of the Chinese Center for Disease Control and Prevention (China CDC, 中国疾病预防控制中心 ).
The CDC itself had only been formed in early 2002 and had to immediately ride through the SARS epidemic. In June 2003, mainland China had no more new or active SARS cases and the SARS epidemic was officially declared as being over. The Institute of Virology was then designated by the Ministry of Health as one of the six storage units for the SARS virus. [b.1]
The Institute of Virology was effectively the most prestigious viral research institution in the country at the time. It had the first P3 in China, was the workplace of famous academicians and was part of the newly formed CDC, which very purpose was to prevent epidemics in the country.

Situated at №100 Yingxin Street, Xicheng District, the institute is 1km from the busy interchange station of Beijing metro lines 4 and 7. The main body of the virus institute was a five-story building (since rebuilt), the fifth floor was a dormitory for staff or visitors , and the remaining four floors were offices and laboratories [b.1].
From the appearance, the whole building was unremarkable. An old man who lived in the bungalow opposite the virus institute for more than 30 years told reporters that the virus centre was built in the 1950s [b.1].
To this day the building is still standing. The Institute main buildings are now at 155 Changbai Rd, Changping Qu, Beijing. But the old building is still part of the Institute. [b.517]
‘№100 Yingxin Street, Beijing still stood stubbornly in that old crowded alley. The windows of the building were covered with grey dust, many were broken, and the exhaust vents jutting from the laboratory pointed at the residents’ homes only a few meters apart.’ [b.3]

Main entrance of the Institute, 100 Yingxin Street — 2020

Side alleys — source: Baidu
At the time there was no logo of the Institute of Virology at the front of the office building [b.1]. Its nondescript appearance clearly contrasted with its flagship role in viral research.

5-storey building of the Institute where the leak happened, source: Baidu
3.3 An academic ego got hurt
The director of the Diarrhoea Virus Department was Academician Hong Tao (洪涛) of the Chinese Academy of Engineering. 72 y-old at the time and a highly respected virus expert, he was one of the main pioneers in the field of medical electron microscopy, the main founder of the virus morphology discipline in China, the discoverer of diarrhoea rotavirus (“洪氏腹泻” Hong’s Diarrhoea) which caused a acute diarrhoea outbreak in North China in 1983, the discoverer of the intracellular morphology of epidemic hemorrhagic fever virus, and one of the founders of the Chinese Society of Electron Microscopy which he used extensively in his virus morphology work. He also had extensively studied chlamydia. [b.11]

Academician Hong Tao, 72 at the time
However recently his prestige had got rather badly damaged.
At the beginning of the 2003 SARS epidemic that started in Guangdong province, on the 13th Feb 2003 precisely, Guangzhou Nanfang Hospital (on the SARS battlefront) issued an autopsy report of a SARS victim, stating that the cause of death was “viral pneumonia.” [b200]
At that very same time Hong Tao and an assistant at the Institute of Virology in Beijing were finding chlamydia particles from lungs tissues of two SARS victims, through electron microscopy.
On the 18th Feb the Institute of Virology of the Chinese Center for Disease Research announced that the main pathogen of atypical pneumonia in Guangdong was likely to be chlamydia. [b.203, b.103]
In the afternoon of the same day, the Guangdong Provincial Department of Health held an emergency meeting. The expert group headed by Zhong Nanshan, a member of the Chinese Academy of Engineering (whose attitude and work during SARS would be exemplary) believed that chlamydia cannot be simply identified as the only pathogen and railed against the limitations of the electron microscope approach.
In effect Hong Tao had made a basic mistake by ignoring the golden rule of infectious disease pathogen identification: Koch’s postulates. More precisely he actually ignored 3 of the 4 Koch’s postulates — quite a feat for an academician. [b501]
“If it is chlamydia, I will cut off my head and give it to you… .”
-- Dr Cai Weiping of the Guangdong expert group , from his hospital bed, after being infected while treating SARS patients [b102]
Both Hong Tao and Zhong Nanshan were members of the elite Chinese Academy of Engineering, but Hong Tao was more senior, based in Beijing, had been in the CCP army back in 49 (very important for connections) and was working for the CDC not for some hospital in the province. Hence the Chlamydia hypothesis became the official theory in China for a while, and totally ineffective antibiotics treatments were formulated accordingly.
While the world scientific community raced ahead, Chinese researchers who disagreed with Dr. Hong couldn’t get their hands on tissue, blood and serum samples from SARS patients.
On March 17–18 the CDC and the Ministry of Health held a first national joint meeting of SARS experts. At that meeting China’s CDC was designated as the country’s leading institution for SARS scientific research, taking the lead back forcefully:
“No unit or individual is allowed to publish research results in any form, including articles; if you want to publish, you must first report to the China CDC.” [b200]
Incapacitated by that incorrect official theory, Chinese scientists could not publish a single descent paper on the origins of SARS at that time [b301]. In particular it would later emerge that:
• Nanfang Hospital (Guangzhou) had found virus particles in lung tissue samples of SARS patients through electron microscopy as early as February 20, but their research was forced to stop after the “ban” issued at the meeting of the 17–18 March [b200]
• Researchers from the Chinese Academy of Military Medical Sciences (Beijing) also discovered virus particles in samples on February 26, which were identified as a coronavirus after being reviewed by 6 virus morphologists. On March 21, the Military Medical College reported to the relevant departments the discovery of the coronavirus. However, the findings were not officially announced until April 9. [b200, b103]
Eventually the Chlamydia theory was proven wrong by scientists abroad (mostly US and HK), by the beginning of April it was clear that a coronavirus was the cause of SARS.
Defying that trend on the 1st Apr 2003, Hong Tao and 20 other Chinese scientists published a paper defending the Chlamydia hypothesis [b207]. Nevertheless on the 3rd April 2003 the WHO declared SARS to be likely caused by a coronavirus [b.510], and on the 16th April 2003 it confirmed it to be the cause. [b509]
Even after the 16th April, an undeterred Hong Tao would tell reporters:
“It does not mean that this virus is the only culprit. [..] We cannot be sure that chlamydia is an important cause of death for patients, but according to our research on previous cases, the coronavirus alone is not so serious.” [b.203]

Academician Zhong Nanshan, 66 at the time