A Proposed Origin for SARS-CoV-2 and the COVID-19 Pandemic [W/Comments]
by Jonathan Latham, PhD and Allison Wilson, PhD
Independent Science News for Food and Agriculture
July 15, 2020
NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHT
YOU ARE REQUIRED TO READ THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
Our supposition as to why there was a time lag between sample collection (in 2012/2013) and the COVID-19 outbreak is that the researchers were awaiting BSL-4 lab construction and certification, which was underway in 2013 but delayed until 2018.
We propose that, when frozen samples derived from the miners were eventually opened in the Wuhan lab they were already highly adapted to humans to an extent possibly not anticipated by the researchers. One small mistake or mechanical breakdown could have led directly to the first human infection in late 2019.
Thus, one of the miners, most likely patient 3, or patient 4 (whose thymus was removed), was effectively patient zero of the COVID-19 epidemic. In this scenario, COVID-19 is not an engineered virus; but, equally, if it had not been taken to Wuhan and no further molecular research had been performed or planned for it then the virus would have died out from natural causes, rather than escaped to initiate the COVID-19 pandemic.
-- A Proposed Origin for SARS-CoV-2 and the COVID-19 Pandemic, by Jonathan Latham, PhD and Allison Wilson, PhD
In all the discussions of the origin of the COVID-19 pandemic, enormous scientific attention has been paid to the molecular character of the SARS-CoV-2 virus, including its novel genome sequence in comparison with its near relatives. In stark contrast, virtually no attention has been paid to the physical provenance of those nearest genetic relatives, its presumptive ancestors, which are two viral sequences named BtCoV/4991 and RaTG13.
This neglect is surprising because their provenance is more than interesting. BtCoV/4991 and RaTG13 were collected from a mineshaft in Yunnan province, China, in 2012/2013 by researchers from the lab of Zheng-li Shi at the Wuhan Institute of Virology (WIV). Very shortly before, in the spring of 2012, six miners working in the mine had contracted a mysterious illness and three of them had died (Wu et al., 2014). The specifics of this mystery disease have been virtually forgotten; however, they are described in a Chinese Master’s thesis written in 2013 by a doctor who supervised their treatment.
We arranged to have this Master’s thesis translated into English. The evidence it contains has led us to reconsider everything we thought we knew about the origins of the COVID-19 pandemic. It has also led us to theorise a plausible route by which an apparently isolated disease outbreak in a mine in 2012 led to a global pandemic in 2019.
The origin of SARS-CoV-2 that we propose below is based on the case histories of these miners and their hospital treatment. This simple theory accounts for all the key features of the novel SARS-CoV-2 virus, including ones that have puzzled virologists since the outbreak began.
The theory can account for the origin of the polybasic furin cleavage site, which is a region of the viral spike protein that makes it susceptible to cleavage by the host enzyme furin and which greatly enhances viral spread in the body. This furin site is novel to SARS-CoV-2 compared to its near relatives (Coutard, et al., 2020). The theory also explains the exceptional affinity of the virus spike protein for human receptors, which has also surprised virologists (Letko et al., 2020; Piplani et al, 2020; Wrapp et al., 2020; Walls et al., 2020). The theory further explains why the virus has barely evolved since the pandemic began, which is also a deeply puzzling aspect of a virus supposedly new to humans (Zhan et al., 2020; van Dorp et al., 2020; Chaw et al., 2020). Lastly, the theory neatly explains why SARS-CoV-2 targets the lungs, which is unusual for a coronavirus (Huang et al., 2020).
We do not propose a specifically genetically engineered or biowarfare origin for the virus but the theory does propose an essential causative role in the pandemic for scientific research carried out by the laboratory of Zheng-li Shi at the WIV; thus also explaining Wuhan as the location of the epicentre.
Why has the provenance of RaTG13 and BtCoV/4991 been ignored?
The apparent origin of the COVID-19 pandemic is the city of Wuhan in Hubei province, China. Wuhan is also home to the world’s leading research centre for bat coronaviruses. There are two virology labs in the city, both have either collected bat coronaviruses or researched them in the recent past. The Shi lab, which collected BtCoV/4991 and RaTG13, recently received grants to evaluate by experiment the potential for pandemic pathogenicity of the novel bat coronaviruses they collected from the wild.
To add to these suggestive data points, there is a long history of accidents, disease outbreaks, and even pandemics resulting from lab accidents with viruses (Furmanski, 2014; Weiss et al., 2015).
This paper presents an historical review of outbreaks of PPPs [Potentially Pandemic Pathogens] or similarly transmissible pathogens that occurred from presumably well-funded and supervised nationally supported laboratories. It should be emphasized that these examples are only the “tip of the iceberg” because they represent laboratory accidents that have actually caused illness outside of the laboratory in the general public environment. The list of laboratory workers who have contracted potentially contagious infections in microbiology labs but did not start community outbreaks is much, much longer. The examples here are not “near misses;” these escapes caused real-world outbreaks.
-- Laboratory Escapes and “Self-fulfilling prophecy” Epidemics, by Martin Furmanski MD, Scientist’s Working Group on Chemical and Biologic Weapons, Center for Arms Control and Nonproliferation, February 17, 2014
For these and other reasons, summarised in our article The Case is Building that COVID-19 Had a Lab Origin, we (a virologist and a geneticist) and others have concluded that a lab outbreak is a credible thesis. Certainly, a lab origin has at least as much circumstantial evidence to support it as does any natural zoonotic origin theory (Piplani et al., 2020; Segreto and Deigin, 2020; Zhan et al., 2020).
Executive Summary
Biological threats—natural, intentional, or accidental—in any country can pose risks to global health, international security, and the worldwide economy. Because infectious diseases know no borders, all countries must prioritize and exercise the capabilities required to prevent, detect, and rapidly respond to public health emergencies. Every country also must be transparent about its capabilities to assure neighbors it can stop an outbreak from becoming an international catastrophe. In turn, global leaders and international organizations bear a collective responsibility for developing and maintaining robust global capability to counter infectious disease threats. This capability includes ensuring that financing is available to fill gaps in epidemic and pandemic preparedness. These steps will save lives and achieve a safer and more secure world.
The Global Health Security (GHS) Index is the first comprehensive assessment and benchmarking of health security and related capabilities across the 195 countries that make up the States Parties1 to the International Health Regulations (IHR [2005]).2 The GHS Index is a project of the Nuclear Threat Initiative (NTI) and the Johns Hopkins Center for Health Security (JHU) and was developed with The Economist Intelligence Unit (EIU). These organizations believe that, over time, the GHS Index will spur measurable changes in national health security and improve international capability to address one of the world’s most omnipresent risks: infectious disease outbreaks that can lead to international epidemics and pandemics.
The GHS Index is intended to be a key resource in the face of increasing risks of high-consequence3 and globally catastrophic4 biological events and in light of major gaps in international financing for preparedness. These risks are magnified by a rapidly changing and interconnected world; increasing political instability; urbanization; climate change; and rapid technology advances that make it easier, cheaper, and faster to create and engineer pathogens.
Developed with the guidance of an international expert advisory panel, the GHS Index data are drawn from publicly available data sources from individual countries and international organizations, as well as an array of additional sources including published governmental information, data from the World Health Organization (WHO), the World Organisation for Animal Health (OIE), the Food and Agriculture Organization of the United Nations (FAO), the World Bank, country legislation and regulations, and academic resources and publications. Unique in the field, the GHS Index provides a comprehensive assessment of countries’ health security and considers the broader context for biological risks within each country, including a country’s geopolitical considerations and health system and whether it has tested its capacities to contain outbreaks.
Knowing the risks, however, is not enough. Political will is needed to protect people from the consequences of epidemics, to take action to save lives, and to build a safer and more secure world.
WHY IS THE GHS INDEX NEEDED?
It is likely that the world will continue to face outbreaks that most countries are ill positioned to combat. In addition to climate change and urbanization, international mass displacement and migration—now happening in nearly every corner of the world—create ideal conditions for the emergence and spread of pathogens. Countries also face an increased potential threat of accidental or deliberate release of a deadly engineered pathogen, which could cause even greater harm than a naturally occurring pandemic. The same scientific advances that help fight epidemic disease also have allowed pathogens to be engineered or recreated in laboratories. Meanwhile, disparities in capacity and inattention to biological threats among some leaders have exacerbated preparedness gaps. The GHS Index seeks to illuminate those gaps to increase both political will and financing to fill them at the national and international levels. Unfortunately, political will for accelerating health security is caught in a perpetual cycle of panic and neglect. Over the past two decades, decision makers have only sporadically focused on health security, despite concerns stemming from the 2001 anthrax attacks, the emergence of the Severe Acute Respiratory Syndrome and Middle East Respiratory Syndrome coronaviruses, and the looming threat of a pandemic caused by a novel strain of influenza.
In September 2014, the United Nations (UN) Security Council met in crisis over the growing Ebola epidemic in West Africa. Massive global assistance was needed to stop the outbreak because of insufficient national capacities in Guinea, Liberia, and Sierra Leone to quickly detect and respond to the epidemic.
As a result, the West Africa Ebola epidemic killed at least 10,000 people and infected more than 28,000.5 The three affected countries lost $2.8 billion in combined GDP, and a massive global response totaled billions of dollars before the outbreak was contained. The crisis awakened the world to the reality that pathogens can emerge unexpectedly, and when outbreaks occur in countries that are unprepared, they can spill beyond borders, threatening the peace, health, and prosperity of all countries. However, despite newly available vaccines and therapies, response to the Ebola outbreak that began in 2018 in eastern Democratic Republic of Congo has been hampered by violence and instability, community resistance to outbreak mitigation measures, hospital transmission, delays in detection and isolation, and lack of funding and resources.
Delays in the global response to Ebola in 2014 led to a restructuring of the WHO and prompted calls for measurement and transparent reporting of countries’ public health capacities, including the launch of the voluntary WHO IHR Joint External Evaluations (JEEs). Since then, health, policy, and security leaders have developed numerous high-level reviews and recommended ways to identify, finance, and fill major preparedness gaps. These recommendations are relevant for epidemic threats, such as Ebola, and high-consequence pandemic threats, such as a fast-spreading respiratory disease agent that could have a geographic scope, severity, or societal impact and could overwhelm national or international capacity to manage it.6 Some of those recommendations have been implemented, but many have been shelved owing in part to lack of financing. Nearly all recommendations pointed to a need to better understand and measure—on a transparent, global, and recurring basis—the state of international capability for preventing, detecting, and rapidly responding to epidemic and pandemic threats.
The GHS Index is designed to meet this need.
DEVELOPING THE GHS INDEX
The NTI, JHU, and EIU project team—with generous grants from the Open Philanthropy Project, the Bill & Melinda Gates Foundation, and the Robertson Foundation—worked with an international advisory panel of 21 experts from 13 countries to create a detailed and comprehensive framework of 140 questions, organized across 6 categories, 34 indicators, and 85 subindicators to assess a country’s capability to prevent and mitigate epidemics and pandemics.
The GHS Index relies entirely on open-source information: data that a country has published on its own or has reported to or been reported by an international entity. The GHS Index was created in this way with a firm belief that all countries are safer and more secure when their populations are able to access information about their country’s existing capacities and plans and when countries understand each other’s gaps in epidemic and pandemic preparedness so they can take concrete steps to finance and fill them. The indicators and questions that compose the GHS Index framework also prioritize analysis of health security capacity in the context of a country’s broader national health system and other national risk factors.
The 140 GHS Index questions are organized across six categories:
1. PREVENTION: Prevention of the emergence or release of pathogens
2. DETECTION AND REPORTING: Early detection and reporting for epidemics of potential international concern
3. RAPID RESPONSE: Rapid response to and mitigation of the spread of an epidemic
4. HEALTH SYSTEM: Sufficient and robust health system to treat the sick and protect health workers
5. COMPLIANCE WITH INTERNATIONAL NORMS: Commitments to improving national capacity, financing plans to address gaps, and adhering to global norms
6. RISK ENVIRONMENT: Overall risk environment and country vulnerability to biological threats
Among its 140 questions, the GHS Index prioritizes not only countries’ capacities, but also the existence of functional, tested, proven capabilities for stopping outbreaks at the source. Several questions in the GHS Index are designed to determine not only whether a capacity exists, but also whether that capacity is regularly—for example, annually—tested and shown to be functional in exercises or real-world events.
The GHS Index also includes indicators of nations’ capacities and capabilities to reduce Global Catastrophic Biological Risks (GCBRs), which are biological risks of unprecedented scale that could cause severe damage to human civilization at a global level, potentially undermining civilization’s long-term potential.7 These are events that could wipe out gains in sustainable development and global health because of their potential to cause national and regional instability, global economic consequences, and widespread morbidity and mortality.
FINDINGS AND RECOMMENDATIONS
This report summarizes the results of the first GHS Index, including overall findings about the state of national health security capacity across each of the six GHS Index categories, as well as additional findings specific to functional areas of epidemic and pandemic preparedness. The full report also offers 33 recommendations to address gaps identified by the GHS Index. All the findings and recommendations are summarized on pages 12–15 and described in detail throughout the full report, which begins on page 31.
Whereas every country has a responsibility to understand, track, improve, and sustain national health security, new and increased global biological risks may require approaches that are beyond the control of individual governments and will necessitate international action. Therefore, the recommendations contained in this report are made with the understanding that health security is a collective responsibility, and a robust international health security architecture is required to support countries at increased risk. As a result, in addition to the many recommendations intended for national leaders, the GHS Index also includes recommendations aimed at decision makers within the UN system, international organizations, donor governments, philanthropies, and the private sector. These are especially important in the case of fast-spreading, deliberately caused, or otherwise unusual outbreaks that could rapidly overwhelm the capability of national governments and international responders.
OVERALL FINDING: National health security is fundamentally weak around the world. No country is fully prepared for epidemics or pandemics, and every country has important gaps to address.
The GHS Index analysis finds no country is fully prepared for epidemics or pandemics. Collectively, international preparedness is weak. Many countries do not show evidence of the health security capacities and capabilities that are needed to prevent, detect, and respond to significant infectious disease outbreaks. The average overall GHS Index score among all 195 countries assessed is 40.2 of a possible score of 100. Among the 60 high-income countries, the average GHS Index score is 51.9. In addition, 116 high- and middle-income countries do not score above 50. Overall, the GHS Index finds severe weaknesses in country abilities to prevent, detect, and respond to health emergencies; severe gaps in health systems; vulnerabilities to political, socioeconomic, and environmental risks that can confound outbreak preparedness and response; and a lack of adherence to international norms.
Specific scores for the GHS Index categories are as follows:
PREVENTION: Fewer than 7% of countries score in the highest tier8 for the ability to prevent the emergence or release of pathogens.
DETECTION AND REPORTING: Only 19% of countries receive top marks for detection and reporting.
RAPID RESPONSE: Fewer than 5% of countries scored in the highest tier for their ability to rapidly respond to and mitigate the spread of an epidemic.
HEALTH SYSTEM: The average score for health system indicators is 26.4 of 100, making it the lowest-scoring category.
COMPLIANCE WITH INTERNATIONAL NORMS: Less than half of countries have submitted Confidence-Building Measures under the Biological Weapons Convention (BWC) in the past three years, an indication of their ability to adhere to important international norms and commitments related to biological threats.
RISK ENVIRONMENT: Only 23% of countries score in the top tier for indicators related to their political system and government effectiveness.
_______________
Notes:
1. As of April 16, 2013, there are 196 States Parties to the World Health Organization (WHO) 2005 International Health Regulations (IHR), including the Holy See. The Holy See is a sovereign juridical entity under international law, but it was not included in the country-specific research for this Index in light of the Holy See’s lack of an independent health system. This report will refer to the assessed “States Parties” as “195 countries.”
2. The WHO IHR (2005) is the foundational international standards for health. The IHR (2005) is a binding legal instrument to address cross-border public health risks. The goal of the IHR (2005) is to prevent, protect, control, and respond without disrupting international trade and traffic. The IHR (2005) provided the guiding regulations behind many of the indicators included in the GHS Index.
3. High-consequence biological events are defined here as infectious disease outbreaks that could overwhelm national or international capacity to manage them. For example, although international health security has improved following the 2014–2016 Ebola epidemic in West Africa, countries and international responders are not prepared to quell outbreaks that occur in violent or insecure settings; deliberate biological events that require close coordination and investigative links between security, health, and humanitarian actors; and fast-moving respiratory diseases with high mortality that could spread rapidly to become global pandemics.
4. Global Catastrophic Biological Risks are biological risks of unprecedented scale that could cause severe damage to human civilization at a global level, potentially undermining its long-term potential. See Nick Alexopoulos, “Center for Health Security Publishes First Working Definition of Global Catastrophic Biological Risks,” Johns Hopkins Center for Health Security, July 27, 2017, http://www.centerforhealthsecurity.org/ ... ition.html.
5. Centers for Disease Control and Prevention, “2014–2016 Ebola Outbreak in West Africa,” http://www.cdc.gov/vhf/ebola/history/20 ... index.html.
6 United Nations General Assembly, “Protecting humanity from future health crises: Report of the High-level Panel on the Global Response to Health Crises,” https://www.un.org/ga/search/view_doc.a ... l=A/70/723.
7. Monica Schoch-Spana et al.,“Global Catastrophic Biological Risks: Toward a Working Definition,” Health Security 15, no. 4 (2017): 323–28, http://www.liebertpub.com/doi/full/10.1089/hs.2017.0038.
8 The GHS Index scoring system includes three tiers. Countries that score between 0 and 33.3 are in the bottom tier (also called “low scores”), countries that score between 33.4 and 66.6 are in the middle tier (also called “moderate scores”), and countries that score between 66.7 and 100 are in the upper or “top” tier (also called “high scores”)
-- 2019 Global Health Security Index: Building Collective Action and Accountability, by Nuclear Threat Initiative, Center for Health Security, Johns Hopkins Bloomberg School of Public Health, and The Economist Intelligence Unit
The media, normally so enamoured of controversy, has largely declined even to debate the possibility of a laboratory escape. Many news sites have simply labelled it a conspiracy theory.
For decades, Dr. Daniel R. Lucey, an infectious disease specialist at Georgetown University, has crisscrossed the globe to study epidemics and their origins. His attention now is on the Covid-19 pandemic, which first came to public notice late last year in Wuhan, China. Its exact beginnings are sufficiently clouded that the World Health Organization has begun a wide inquiry into its roots. The advance team is to leave for China this weekend, and Dr. Lucey has publicly encouraged the health agency to address what he considers eight top questions....
The sixth and seventh questions go to whether the deadly pathogen leapt to humans from a laboratory. Although some intelligence analysts and scientists have entertained that scenario, no direct evidence has come to light suggesting that the coronavirus escaped from one of Wuhan’s labs.
Even so, given the wet market’s downgrading in the investigation, “It is important to address questions about any potential laboratory source of the virus, whether in Wuhan or elsewhere,” Dr. Lucey wrote in his blog post.
To that end, he urges the W.H.O. investigators to look for any signs of “gain of function” research — the deliberate enhancement of pathogens to make them more dangerous. The technique is highly contentious. Critics question its merits and warn that it could lead to catastrophic lab leaks. Proponents see it as a legitimate way to learn how viruses and other infectious organisms might evolve to infect and kill people, and thus help in devising new protections and precautions.
Debate over its wisdom erupted in 2011 after researchers announced success in making the highly lethal H5N1 strain of avian flu easily transmissible through the air between ferrets, at least in the laboratory.Prompted by controversy over dangerous research and recent laboratory accidents, the White House announced Friday that it would temporarily halt all new funding for experiments that seek to study certain infectious agents by making them more dangerous.
It also encouraged scientists involved in such research on the influenza, SARS and MERS viruses to voluntarily pause their work while its risks were reassessed.
Opponents of this type of research, called gain of function — for example, attempts to create a more contagious version of the lethal H5N1 avian influenza to learn which mutations made it that way — were elated.
“Brilliant!” said Peter Hale, the executive director of the Foundation for Vaccine Research, which opposes such experiments. “The government has finally seen the light. This is what we have all been waiting for and campaigning for. I shall sleep better tonight.”
The announcement, which was made by the White House Office of Science and Technology Policy and the Department of Health and Human Services, did not say how long the moratorium would last. It said a “deliberative process to assess the potential risks and benefits” would begin this month and stretch at least into next year.
The move appeared to be a sudden change of heart by the Obama administration, which last month issued regulations calling for more stringent federal oversight of such research and requiring scientists and universities to disclose that their work might be risky, rather than expecting federal agencies to notice.
Critics at the time dismissed those rules as too weak.
The moratorium is only on research on influenza virus and the coronaviruses that cause SARS and MERS. It made no mention of Ebola or any related filovirus. Ebola is already extremely lethal, but it is not easily transmissible.
No scientist has publicly announced an attempt to make Ebola as easy to transmit with a sneeze as flu is. Given the current panic around Ebola, and congressional anger at federal health agencies, it is unlikely that federal funding for such a project would be given out.
The debate over the wisdom of “gain of function” research erupted in 2011 when the labs of Ron Fouchier of Erasmus University in the Netherlands, and Yoshihiro Kawaoka of the University of Wisconsin-Madison, separately announced that they had succeeded in making the lethal H5N1 avian flu easily transmissible between ferrets, which are a model for human susceptibility to flu.
The debate heated up further this year when the Centers for Disease Control and Prevention admitted it had suffered laboratory accidents that exposed dozens of workers to anthrax and shipped deadly avian flu virus to another federal lab that had asked for a more benign flu strain. Also this year, vials of smallpox that had been forgotten for 50 years were found in a lab at the National Institutes of Health.
The White House said the moratorium decision had been made “following recent biosafety incidents at federal research facilities.”
Dr. Kawaoka said he would not start any new gain-of-function experiments and would consult with the N.I.H. about which ones he had underway that met their criteria for the moratorium.
Many scientists were furious that such work had been permitted and even supported with American tax dollars. But others argued that it was necessary to learn which genetic mutations make viruses more dangerous. If those mutations began appearing naturally as the viruses circulated in animals and people, warnings could be issued and vaccines designed, they said.
Some scientists argued that the two scientists should not be permitted to publish all the details of their experiments, for fear that terrorists or unscrupulous scientists would duplicate them and start a fatal pandemic.
Others, like Richard H. Ebright, a molecular biologist and bioweapons expert at Rutgers University, argued that the long history of accidental releases of infectious agents from research labs made such work extremely risky and unwise to perform in the first place.
Dr. Ebright called Friday’s announcement “an important, albeit overdue, step.”
Michael T. Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, called the moratorium “a wise move — I congratulate the U.S. government on taking this step.”
The new policy had to be announced now, he explained, because the National Science Advisory Board for Biosecurity is to meet later this month. It will have 11 new members, and gain of function research is a principal agenda item.
Dr. Osterholm was one of 11 previous members who were removed from the board in the middle of the controversy.
All, like him, had been on it many years past their original five-year appointments and were due to be replaced, but had routinely been asked to stay, he said.
In April, he was the author of a letter to the National Institutes of Health complaining about government pressure on the advisory board. The institutes gave grants to support gain of function work.
The explanation given was that they had outlasted their tenures, but Dr. Osterholm said that “in the same week as the anthrax accident at the C.D.C., we all got an email on a Sunday night from a junior staffer telling us we were out.”
He called that a public relations failure: “P.R. zero point zero.”
-- White House to Cut Funding for Risky Biological Study, by Donald G. McNeil Jr., NYT, Oct. 17, 2014
In his blog, Dr. Lucey asks “what, if any,” gain-of-function studies were done on coronaviruses in Wuhan, elsewhere in China, or in collaboration with foreign laboratories.
“If done well scientifically, then this investigation should allay persistent concerns about the origin of this virus,” he wrote. “It could also help set an improved standard for investigating and stopping the awful viruses, and other pathogens, in the decades ahead.”
Finally, Dr. Lucey asks the W.H.O. team to learn more about China’s main influenza research lab, a high-security facility in Harbin, the capital of China’s northernmost province. In May, he notes, a Chinese paper in the journal Science reported that two virus samples from Wuhan were studied there in great detail early this year, including in a variety of animals. It reported that cats and ferrets were highly susceptible to the pathogen; dogs were only mildly susceptible; and pigs, chickens and ducks were not susceptible at all.
-- 8 Questions From a Disease Detective on the Pandemic’s Origins: Dr. Daniel R. Lucey wants answers to pointed questions that bear on how the coronavirus leapt from bats to humans, by William J. Broad, NYT, July 8, 2020
The principal reason for media dismissals of the lab origin possibility is a review paper in Nature Medicine (Andersen et al., 2020). Although by Jun 29, 2020 this review had almost 700 citations it also has major scientific shortcomings. These flaws are worth understanding in their own right but they are also useful background for understanding the implications of the Master’s thesis.
An influential paper was published in Nature Medicine on 17 March 2020. Andersen et al observed that several mutations have occurred in the receptor binding domain of SARS-CoV-2. These, they suggested, therefore sustain an hypothesis of natural evolution (Andersen et al., 2020). We do not agree. We do agree that it is indeed correct that several such mutations are to be seen and in a forthcoming companion article to this one, about three other viruses of interest, we will discuss further Andersen et al's evidence and argumentation in that context. But here we observe only that the contention that it is improbable that Covid-19 emerged through laboratory manipulation of a related SARS-CoV-like coronavirus because the ACE2 binding is not ideal is weakened because Andersen et al cite two authorities which actually say the reverse of what they say that they say.
Wan et al are cited by Andersen et al but offer them no support (Wan et al., 2020). Wan et al say, correctly in our view, that computational structural modelling of complex virus-receptor interactions can be used for structural predictions and that such models can potentially be used for Gain-Of-Function modelling. It is well known that models have been developed from data generated in animal model systems such as the palm civet. Wan et al say that the SARS-CoV-2 binding to the ACE2 receptor confirms the accuracy of the structural predictions. Therefore the data and conclusion in Wan et al contradicts Andersen et al's opinion that it is improbable that the virus could have emerged through laboratory manipulation.
There is a similar problem with (Sheahan et al., 2008). This deals with research on a civet strain SZ16 and the infective strain SARS-CoV Urbani. These strains were used to create a chimeric virus icSZ16-S. Sheahan et al go on to explain that by in vitro evolution of the chimeric virus icSZ16-S on human airway epithelial (HAE) cells in the lab, they have been able to produce two new viruses binding to such HAE cells. Therefore this reference supports the very opposite of the Andersen et al hypothesis. We are immediately wary of any paper containing such egregious errors.
-- The Evidence which Suggests that This Is No Naturally Evolved Virus: A Reconstructed Historical Aetiology of the SARS-CoV-2 Spike, by Birger Sørensen, Angus Dalgleish & Andres Susrud
The Nature Paper vs. the Lab-Made Hypothesis
But didn’t that Nature article refute the lab-made hypothesis? No, not really. There is no irrefutable evidence against it in the paper, just a loud “we don’t believe so” based on a shaky foundation. Judge for yourself — here are the authors’ key arguments in support of their conclusions:While the analyses above suggest that SARS-CoV-2 may bind human ACE2 with high affinity, computational analyses predict that the interaction is not ideal and that the RBD sequence is different from those shown in SARS-CoV to be optimal for receptor binding. Thus, the high-affinity binding of the SARS-CoV-2 spike protein to human ACE2 is most likely the result of natural selection on a human or human-like ACE2 that permits another optimal binding solution to arise. This is strong evidence that SARS-CoV-2 is not the product of purposeful manipulation.
In the original paper, the quoted sentences are just below the diagram showing identical RBMs between CoV2 and pangolin-2019. So I am puzzled as to what “computational analysis” has to do with anything. Obviously, the most likely scenario for the lab-made hypothesis is the transfer of RBM from one strain to another — which virologists have done many times before. Therefore, the author’s chain of arguments does not make sense: “computer says binding is not ideal, thus CoV2 must be the result of natural selection. Ergo, this is strong evidence that CoV2 is not lab-made.” Wait, just because CoV2 differs from some “optimal” virus, doesn’t mean it could not have been created in a lab. Not the lab trying to create “optimal” bioweapons, but a lab creating chimeras of naturally found strains, say, in bats and pangolins.
The authors continue to surprise:Furthermore, if genetic manipulation had been performed, one of the several reverse-genetic systems available for betacoronaviruses would probably have been used. However, the genetic data irrefutably show that SARS-CoV-2 is not derived from any previously used virus backbone.
Again, the same questionable logic dressed in categorical adjectives: “genetic analysis irrefutably proves that CoV2 was not created on the basis of previously known strains!” Well thanks, Captain Obvious. But why couldn’t potential creators of CoV2 make a cDNA backbone from unpublished strains related to or even derived from RaTG13? Then they could easily insert the pangolin RBM into it, as well as add a furin site (or maybe the cDNA backbone already had one). Virologists have been doing things like this for 20 years, and modern genetic engineering tools make such manipulations accessible even to a grad student.
As for the chances of the furin site arising in cell culture, the authors also express strange ideas:The acquisition of both the polybasic cleavage site and predicted O-linked glycans also argues against culture-based scenarios. New polybasic cleavage sites have been observed only after prolonged passage of low-pathogenicity avian influenza virus in vitro or in vivo. Furthermore, a hypothetical generation of SARS-CoV-2 by cell culture or animal passage would have required prior isolation of a progenitor virus with very high genetic similarity, which has not been described. Subsequent generation of a polybasic cleavage site would have then required repeated passage in cell culture or animals with ACE2 receptors similar to those of humans, but such work has also not previously been described.
First off, the authors themselves cite previous works where the furin site arose in vitro as viruses were cultured in cells. And second, what do they mean, a strain with high genetic similarity has not been described — what about RaTG13? If it had its RBM replaced by one from the pangolin strain, and then the chimeric strain was cultured in vitro, then the furin site could well have arisen in this matter. Additionally, the new strain could thus acquire other mutations that distinguish CoV2 from RaTG13 and pangolin-2019.
But in terms of the potential lab-based origin of the furin site, I am more inclined to hypothesize a specific insertion — as in the Beijing paper from October 2019 with chicken coronavirus. After that, the synthetic strain could have acquired new mutations by subsequent culturing in vitro or in vivo — like the MA15 murine strain in 2007, for example. Or maybe even using the same mouse model with humanized lung tissues and immune system that was created at UNC by Baric’s and other groups in 2018, in which they reported testing several viruses including MERS:The human innate and adaptive immune system of BLT-L mice
We generated an in vivo model with human lung implants and an autologous human immune system by constructing BLT mice with autologous human lung implants (BLT-L humanized mice).
Finally, even if CoV2 is the product of selection rather than intelligent design, that does not rule out a lab leak either — selection can happen in the lab just as well, both natural and artificial kinds. Different strains can recombine in research animals or in vitro by design or by chance.
-- Lab-Made? SARS-CoV-2 Genealogy Through the Lens of Gain-of-Function Research, by Yuri Deigin
Andersen et al., a critique
The question of the origin of the COVID-19 pandemic is, in outline, simple. There are two incontrovertible facts. One, the disease is caused by a human viral pathogen, SARS-CoV-2, first identified in Wuhan in December 2019 and whose RNA genome sequence is known. Second, all of its nearest known relatives come from bats. Beyond any reasonable doubt SARS-CoV-2 evolved from an ancestral bat virus. The task the Nature Medicine authors set for themselves was to establish the relative merits of each of the various possible routes (lab vs natural) by which a bat coronavirus might have jumped to humans and in the same process have acquired an unusual furin site and a spike protein having very high affinity for the human ACE2 receptor.
When Andersen et al. outline a natural zoonotic pathway they speculate extensively about how the leap might have occurred. In particular they elaborate on a proposed residence in intermediate animals, likely pangolins. For example, “The presence in pangolins of an RBD [Receptor Binding Domain] very similar to that of SARS-CoV-2 means that we can infer that this was probably in the virus that jumped to humans. This leaves the insertion of [a] polybasic cleavage site to occur during human-to-human transmission.” This viral evolution occurred in “Malayan pangolins illegally imported into Guangdong province”. Even with these speculations there are major gaps in this theory. For example, why is the virus so well adapted to humans? Why Wuhan, which is 1,000 Km from Guangdong? (See map).
china province guide
The authors provide no such speculations in favour of the lab accident thesis, only speculation against it:
“Finally, the generation of the predicted O-linked glycans is also unlikely to have occurred due to cell-culture passage, as such features suggest the involvement of an immune system.” (italics added).
[Passaging is the deliberate placing of live viruses into cells or organisms to which they are NOT adapted for the purpose of making them adapted, i.e. speeding up their evolution.]
It is also noteworthy that the Andersen authors set a higher hurdle for the lab thesis than the zoonotic thesis. In their account, the lab thesis is required to explain all of the evolution of SARS-CoV-2 from its presumed bat viral ancestor, whereas under their telling of the zoonotic thesis the key step of the addition of the furin site is allowed to happen in humans and is thus effectively unexplained.
A further imbalance is that key information needed to judge the merits of a lab origin theory is missing from their account. As we detailed in our previous article, in their search for SARS-like viruses with zoonotic spillover potential, researchers at the WIV have passaged live bat viruses in monkey and human cells (Wang et al., 2019). They have also performed many recombinant experiments with diverse bat coronaviruses (Ge et al., 2013; Menachery et al., 2015; Hu et al., 2017). Such experiments have generated international concern over the possible creation of potential pandemic viruses (Lipsitch, 2018). As we showed too, the Shi lab had also won a grant to extend that work to whole live animals. They planned “virus infection experiments across a range of cell cultures from different species and humanized mice” with recombinant bat coronaviruses. Yet Andersen et al did not discuss this research at all, except to say:
“Basic research involving passage of bat SARS-CoV-like coronaviruses in cell culture and/or animal models has been ongoing for many years in biosafety level 2 laboratories across the world”
This statement is fundamentally misleading about the kind of research performed at the Shi lab.
A further important oversight by the Andersen authors concerns the history of lab outbreaks of viral pathogens. They write: “there are documented instances of laboratory escapes of SARS-CoV”. This is a rather matter-of-fact allusion to the fact that since 2003 there have been six documented outbreaks of SARS from labs, not all in China, with some leading to fatalities (Furmanski, 2014).
Andersen et al might have also have noted that two major human pandemics are widely accepted to have been caused by lab outbreaks of viral pathogens, H1N1 in 1977 and Venezuelan Equine Encephalitis (summarised in Furmanski, 2014). Andersen could even have noted that literally hundreds of lab accidents with viruses have resulted in near-misses or very localised outbreaks (summarised by Lynn Klotz and Sam Husseini and also Weiss et al., 2015).
Also unmentioned were instances where a lab outbreak of an experimental or engineered virus has been plausibly theorised but remains uninvestigated. For example, the most coherent explanation for the H1N1 variant ‘swine flu’ pandemic of 2009/10 that resulted in a death toll estimated by some as high as 200,000 (Duggal et al., 2016; Simonsen et al. 2013), is that a vaccine was improperly inactivated by its maker (Gibbs et al., 2009). If so, H1N1 emerged from a lab not once but twice.
Given that human and livestock viral outbreaks have frequently come from laboratories and that many scientists have warned of probable lab escapes (Lipsitch and Galvani, 2014), and that the WIV [Wuhan Institute of Virology] itself has a questionable biosafety record, the Andersen paper is not an even-handed treatment of the possible origins of the COVID-19 virus.
This chapter makes the case against performing exceptionally dangerous gain-of-function experiments that are designed to create potentially pandemic and novel strains of influenza, for example, by enhancing the airborne transmissibility in mammals of highly virulent avian influenza strains. This is a question of intense debate over the last 5 years, though the history of such experiments goes back at least to the synthesis of viable influenza A H1N1 (1918) based on material preserved from the 1918 pandemic. This chapter makes the case that experiments to create potential pandemic pathogens (PPPs) are nearly unique in that they present biosafety risks that extend well beyond the experimenter or laboratory performing them; an accidental release could, as the name suggests, lead to global spread of a virulent virus, a biosafety incident on a scale never before seen. In such cases, biosafety considerations should be uppermost in the consideration of alternative approaches to experimental objectives and design, rather than being settled after the fact, as is appropriately done for most research involving pathogens. The extensive recent discussion of the magnitude of risks from such experiments is briefly reviewed. The chapter argues that, while there are indisputably certain questions that can be answered only by gain-of-function experiments in highly pathogenic strains, these questions are narrow and unlikely to meaningfully advance public health goals such as vaccine production and pandemic prediction. Alternative approaches to experimental influenza virology and characterization of existing strains are in general completely safe, higher throughput, more generalizable, and less costly than creation of PPP in the laboratory and can thereby better inform public health. Indeed, virtually every finding of recent PPP experiments that has been cited for its public health value was predated by similar findings using safe methodologies. The chapter concludes that the unique scientific and public health value of PPP experiments is inadequate to justify the unique risks they entail and that researchers would be well-advised to turn their talents to other methodologies that will be safe and more rewarding scientifically.
-- Why Do Exceptionally Dangerous Gain-of-Function Experiments in Influenza?, by Marc Lipsitch
Yet its text expresses some strong opinions: “Our analyses clearly show that SARS-CoV-2 is not a laboratory construct or a purposefully manipulated virus….It is improbable that SARS-CoV-2 emerged through laboratory manipulation of a related SARS-CoV-like coronavirus…..the genetic data irrefutably show that SARS-CoV-2 is not derived from any previously used backbone….the evidence shows that SARS-CoV2 is not a purposefully manipulated virus….we do not believe that any type of laboratory-based scenario is possible.” (Andersen et al., 2020).
It is hard not to conclude that what their paper mostly shows is that Drs. Andersen, Rambaut, Lipkin, Holmes and Garry much prefer the natural zoonotic transfer thesis. Their rhetoric is forthright but the evidence does not support that confidence.
Indeed, since the publication of Andersen et al., important new evidence has emerged that undermines their zoonotic origin theory. On May 26th the Chinese CDC ruled out the Huanan “wet” market in Wuhan as the source of the outbreak. Additionally, new research on pangolins, the favoured intermediate mammal host, suggests they are not a natural reservoir of coronaviruses (Lee et al., 2020; Chan and Zhan, 2020). Furthermore, SARS-CoV-2 was found not to replicate in bat kidney or lung cells (Rhinolophus sinicus), implying that SARS-CoV-2 is not a recently-adapted spill over Chu et al., 2020).
The Mojiang mine and the Master’s thesis
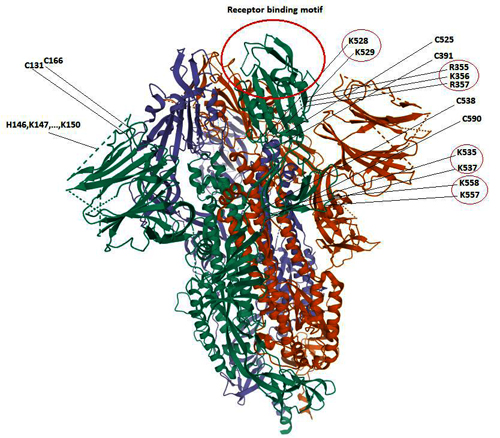
In our own search to resolve the COVID-19 origin question we chose to focus on the provenance of the coronavirus genome sequences BtCoV/4991 and RaTG13, since these are the most closely related sequences to SARS-CoV-2 (98.7% and 96.2% identical respectively). See FIG 1. (reproduced from P. Zhou et al., 2020).
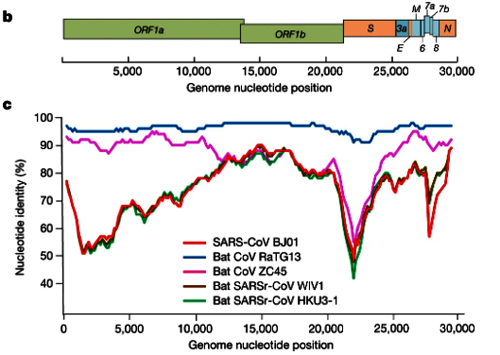
Similarity of SARS-CoV-2 to RaTG13 (blue line) and other coronaviruses (red, green, pink) (Image from Zhou et al., 2020). The higher the line the more similar the virus.
For comparison, the next closest virus to SARS-CoV-2 is RmYN02 (not shown in Fig 1.) (H. Zhou et al., 2020). RmYN02 has an overall similarity to SARS-CoV-2 of 93.2%, making its evolutionary distance from SARS-CoV-2 almost twice as great.
BtCoV/4991 was first described in 2016. It is a 370 nucleotide virus fragment collected from the Mojiang mine in 2013 by the lab of Zeng-li Shi at the WIV [Wuhan Institute of Virology] (Ge et al., 2016). BtCoV/4991 is 100% identical in sequence to one segment of RaTG13. RaTG13 is a complete viral genome sequence (almost 30,000 nucleotides) that was only published in 2020, after the pandemic began (P. Zhou et al., 2020).
Despite the confusion created by their different names, in a letter obtained by us Zheng-li Shi confirmed to a virology database that BtCoV/4991 and RaTG13 are both from the same bat faecal sample and the same mine. They are thus sequences from the same virus. In the discussion below we will refer primarily to RaTG13 and specify BtCoV/4991 only as necessary.
These specifics are important because it is these samples and their provenance that we believe are ultimately key to unravelling the mystery of the origins of COVID-19.
The story begins in April 2012 when six workers in that same Mojiang mine fell ill from a mystery illness while removing bat faeces. Three of the six subsequently died.
In a March 2020 interview with Scientific American Zeng-li Shi dismissed the significance of these deaths, claiming the miners died of fungal infections. Indeed, no miners or deaths are mentioned in the paper published by the Shi lab documenting the collection of RaTG13 (Ge et al., 2016).
But Shi’s assessment does not tally with any other contemporaneous accounts of the miners and their illness (Rahalkar and Bahulikar, 2020). As these authors have pointed out, Science magazine wrote up part of the incident in 2014 as A New Killer Virus in China?. Science was citing a different team of virologists who found a paramyxovirus in rats from the mine. These virologists told Science they found “no direct relationship between human infection” and their virus. This expedition was later published as the discovery of a new virus called MojV after Mojiang, the locality of the mine (Wu et al., 2014).
What this episode suggests though is that these researchers were looking for a potentially lethal virus and not a lethal fungus. Also searching the Mojiang mine for a [potentially lethal] virus at around the same time was Canping Huang, the author of a PhD thesis carried out under the supervision of George Gao, the head of the Chinese CDC.
All of this begs the question of why the Shi lab, which has no interest in fungi but a great interest in SARS-like bat coronaviruses, also searched the Mojiang mine for bat viruses on four separate occasions between August 2012 and July 2013, even though the mine is a 1,000 Km from Wuhan (Ge et al., 2016). These collecting trips began while some of the miners were still hospitalised.
Fortunately, a detailed account of the miner’s diagnoses and treatments exists. It is found in a Master’s thesis written in Chinese in May 2013. Its suggestive English title is “The Analysis of 6 Patients with Severe Pneumonia Caused by Unknown viruses“.
The original English version of the abstract implicates a SARS-like coronavirus as the probable causative agent and that the mine “had a lot of bats and bats’ feces”.
The findings of the Master’s thesis
To learn more, especially about the reasonableness of this diagnosis, we arranged to have the whole Master’s thesis translated into English and are here making the translation available. To read it in full see the embedded document below (or download it here).
[x]
Master's Thesis: "The Analysis of Six Patients With Severe Pneumonia Caused By Unknown Viruses"
The six ill miners were admitted to the No. 1. School of Clinical Medicine, Kunming Medical University, in short succession in late April and early May 2012. Kunming is the capital of Yunnan province and 250 Km from Mojiang.
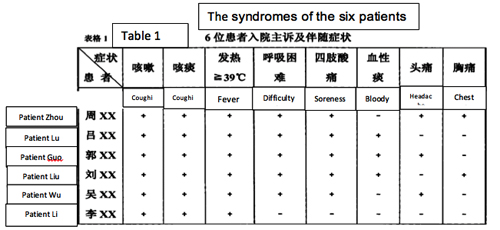
The Syndromes of the six Mojiang Mine patients
Of the descriptions of the miners and their treatments, which include radiographs and numerous CAT scans, several features stand out:
1) From their admission to the hospital their doctors informed the “medical office” of a potential “outburst of disease” i.e. a potential epidemic outbreak. Thus, the miners were treated for infections and not as if they had inhaled noxious gases or other toxins.
2) The symptoms (on admission) of the six miners were: a) dry cough, b) sputum, c) high fevers, especially shortly before death d) difficulty breathing, e) myalgia (sore limbs). Some patients had hiccoughs and headaches. (See Table 1).
3) Clinical work established that patients 1-4 had low blood oxygen “for sure it was ARDS” (Acute Respiratory Distress Syndrome) and immune damage considered indicative of viral infection. Additionally, a tendency for thrombosis was noted in patients 2 and 4. Symptom severity and mortality were age-related (though from a sample of 6 this must be considered anecdotal).
4) Potential common and rare causes of their symptoms were tested for and mostly eliminated. For patients 3 and 4 these included tests for HIV, Cytomegalovirus, Epstein-Barr Virus (EBV), Japanese encephalitis, haemorrhagic fever, Dengue, Hepatitis B, SARS, and influenza. Of these, only patient 2 tested positive for Hepatitis and EBV.
5) Treatment of the six patients included ventilation (patients 2-4), steroids (all patients), antivirals (all except patient 5), and blood thinners (patients 2 and 4). Antibiotics and antifungal medications were administered to counter what were considered secondary (but significant) co-infections.
6) A small number of remote meetings were held with researchers at other universities. One was with Zhong Nanshan at Sun Yat-Sen University, Guangdong. Zhong is the Chinese hero of the SARS epidemic, a virologist, and arguably the most famous scientist in China.
7) Samples from the miners were later sent to the WIV in Wuhan and to Zhong Nanshan, further confirming that viral disease was strongly suspected. Some miners did test positive for coronavirus (the thesis is unclear on how many).
8) The source of infection was concluded to be Rhinolophus sinicus, a horseshoe bat and the ultimate conclusion of the thesis reads “the unknown virus lead to severe pneumonia could be: The SARS-like-CoV from the Chinese rufous horseshoe bat.” Thus the miners had a coronavirus but it apparently was not SARS itself.