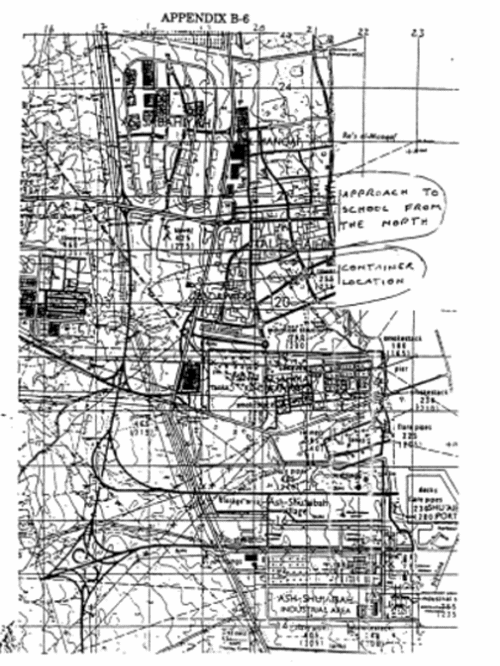
United States Dual-Use Exports to Iraq and Their Impact
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-8
6225. Mustard Gas. 1,1' -Thiobis[2-chloroethane]; bis- (2-chloroethyl)sulfide: B,B'-dichlorocthyl sulfide: 2,2' -di- chlorodiethyl sulfide; bis(B-chloroethyl)sulfide; 1-chloro-2- (B-chloroethylthio)ethane; sulfur mustard: yellow cross li- quid; Kampfstoff "Lost"; Yperite. C4H8,C12S; mol wt 159.08. C 30.20%, H 5.07%, Cl 44.58%. S 20.16%.. (ClCH2CH2)2S. War gas prepd by treating ethylene with sulfur chloride (Levinstein process): Mann. Pope, J. Chem. Soc. 121, 594 (1922): by treating B,B'-dihydroxyethyl sulfide with HCI gas (German process): Meyer. Ber. 19, 3260 (1886): Ann. 240, 310 (1887): Gomberg, J. Am. Chem. Soc. 41, 1427 (1919). Reactions and derivatives: Helfrich, Reid, Ibid 42, 1208 (1920). Toxicity: Anslow et al., J. Pharmacol Exp. Ther. 93, 1 (1948). Review of carcinogenicity studies: IARC Monographs 9, 181-192 (1975).
Oily liquid. Deadly vesicant. Weak, sweet, agreeable odor. On cooling it forms prisms, mp 13-14°. d13 1.338 (solid); df204 1.2741 (liq). bp760 215-217°; bp10 98°. Volatile with steam. n20D 1.53125. Very sparingly sol in water, sol in fat solvents, other common organic solvents. High lipid soly. Vapor pressure at 0° = 0.025 mm; at 30° = 0.090 mm. Hydrolyzed by alkalies. Recommended neutralizing agent and inactivator: Bleaching powder, sodium hypochlorite. LD50 in rats, mice (mg/kg): 3.3. 8.6 i.v. (Anslow).
Human Toxicity: Conjunctivitis, blindness. Produces delayed effects. In 1-12 hrs cough, edema of eyelids, erythema of skin, severe pruritus. May cause edema, ulceration, necrosis of respiratory tract and exposed skin. Ingestion of contaminated material may cause nausea and vomiting. Permanent eye damage, severe respiratory impairment may result. This substance has been tested as a known carcinogen: Fourth Annual Report on Carcinogens (NTP 85-002, 1985) p 136.
USE: In chemical warfare.
6225. Mustard Gas. 1,1' -Thiobis[2-chloroethane]; bis- (2-chloroethyl)sulfide: B,B'-dichlorocthyl sulfide: 2,2' -di- chlorodiethyl sulfide; bis(B-chloroethyl)sulfide; 1-chloro-2- (B-chloroethylthio)ethane; sulfur mustard: yellow cross li- quid; Kampfstoff "Lost"; Yperite. C4H8,C12S; mol wt 159.08. C 30.20%, H 5.07%, Cl 44.58%. S 20.16%.. (ClCH2CH2)2S. War gas prepd by treating ethylene with sulfur chloride (Levinstein process): Mann. Pope, J. Chem. Soc. 121, 594 (1922): by treating B,B'-dihydroxyethyl sulfide with HCI gas (German process): Meyer. Ber. 19, 3260 (1886): Ann. 240, 310 (1887): Gomberg, J. Am. Chem. Soc. 41, 1427 (1919). Reactions and derivatives: Helfrich, Reid, Ibid 42, 1208 (1920). Toxicity: Anslow et al., J. Pharmacol Exp. Ther. 93, 1 (1948). Review of carcinogenicity studies: IARC Monographs 9, 181-192 (1975).
Oily liquid. Deadly vesicant. Weak, sweet, agreeable odor. On cooling it forms prisms, mp 13-14°. d13 1.338 (solid); df204 1.2741 (liq). bp760 215-217°; bp10 98°. Volatile with steam. n20D 1.53125. Very sparingly sol in water, sol in fat solvents, other common organic solvents. High lipid soly. Vapor pressure at 0° = 0.025 mm; at 30° = 0.090 mm. Hydrolyzed by alkalies. Recommended neutralizing agent and inactivator: Bleaching powder, sodium hypochlorite. LD50 in rats, mice (mg/kg): 3.3. 8.6 i.v. (Anslow).
Human Toxicity: Conjunctivitis, blindness. Produces delayed effects. In 1-12 hrs cough, edema of eyelids, erythema of skin, severe pruritus. May cause edema, ulceration, necrosis of respiratory tract and exposed skin. Ingestion of contaminated material may cause nausea and vomiting. Permanent eye damage, severe respiratory impairment may result. This substance has been tested as a known carcinogen: Fourth Annual Report on Carcinogens (NTP 85-002, 1985) p 136.
USE: In chemical warfare.
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-9
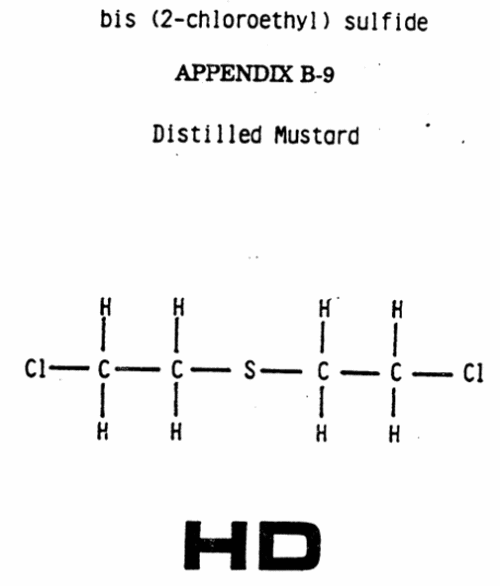
Distilled Mustard
HD
Standard NATO agreement (STANAG) code: HD
Chemical name: bis (2-chloroethyl) sulfide Common name: distilled mustard [1] Formula: C1(CH2)2S(CH2)2C1
Family: casualty agent
Type: blister agent (mustard)
An amber brown liquid with an odor similar to that of burning garlic, the odor becoming more pronounced with impurities in the solution. Creates a lowlaying colorless vapor around the splashed liquid.
History
Distilled mustard (HD) was known as early as the late 1880s as a by-product of the dye industry whose toxic effects might be of use in both treating minor tumors and in killing warehouse pests.
Originally investigated by the British and rejected as not being lethal enough, it was first used against them. At 10 PM on the evening of July 12th, 1917 the British 15th and 55th infantry divisions came under an artillery barrage in their positions near Ypres, France. Mixed in with high explosives was a significant quantity of distilled mustard. Other than a stench "like garlic" of "like mustard" (which is how the compound gets its name) there was little nuisance beyond eye irritation like diluted phosgene. Many troops did not bother to put their gas masks on. By the following afternoon the British field hospitals were clogged and on the next evening the first deaths began.
Distilled mustard, the first of the major blister agents, became the chemical compound by which all others have been judged since. According to a British army study, the Foulkes Papers, 16-1/2% of all encountered casualties in the last eighteen months of that war were due to chemicals, mainly distilled mustard. In 1920 about 19,000 British veterans drew permanent disability, mainly from mustard. A 1927 Porton Down study of HD victims found pre-cardiac conditions common, and typical conjunctivitis, laryngitis, bronchitis, and reoccurring skin burns. A Porton Down study of 1929 of 29 severe cases showed fibrosis, respiratory and spinal TB, persistent laryngitis, anemia, conjunctivitis, aphonia, and pulmonary fibrosis. By 1930, 80% of known survivors suffered chronic bronchitis.
After the first world war, limited production continued in Great Britain and France. Production began in Italy and the Soviet Union by 1925, and in Japan in 1928.
HD was allegedly used against the Afghans by the British in 1919. It was known to be used against Moroccans by the French and Spanish in 1925, against Ethiopians by the Italians after 1935, and by the Japanese against China after 1934 and ending by 1944.
The late 1920s through the 1930s were a time a chemical warfare experimentation as the older agents were adapted to fit new methods and tactics of war. New munitions, especially those for aerial sprays and bombs, were developed. It was assumed that chemicals would be used in the the "next war" to the same extent they had been used in 1915-1918. As German rearmament beginning in 1934 started a general trend, chemical munitions plants were upgraded.
Great Britain built new facilities such as those at Sutton Oak-St Helens, Lancashire. France opened a HD and CG plant at Clancy. The USSR built new HD-L-CG plants at Brandyuzhsky, Kuibyshev, and Karaganda. Germany built plants at Munster, Wunsdorf, and List. The U.S. reluctantly reopened Pine Bluff Arsenal (in production by 1942) and built new facilities such as Rocky Mountain Arsenal (near Denver) and opened the Dugway, Utah, Proving Grounds for the express purpose of testing chemical agents.
Casualty agent use in the second world war, outside of China through 1943-1944, was virtually non-existent. The only known incident in the west occurred in Bari harbor on the Italian Adriatic.
An American merchantman, the S.S. John Harvey, carried the bulk of U.S. chemical munitions for the Mediterranean area, in the form of 2000 M47A1 aerial bombs, containing a total of about 100 tons of HD. The S.S. John Harvey's cargo was known only to SHAFE in London and a few junior Army Ordinance officers on board. On 7:30 PM, July 2nd, 1943, the John Harvey was sunk in a German air raid. Her cargo of HD had all been made by the quick-but-dirty Levinstein process. Thus it had hydrogen and ethane gas impurities in the HD mix. The John Harvey blew up and flooded the harbor. By the following morning it was Ypres all over again, except nobody knew why the massive blistering was happening, especially among sailors soaked in oil and some smelly stuff from the harbor waters. About 630 serious military cases occurred and over a thousand Italian civilians died within a few days.
Allegations of the use of distilled mustard surface from time to time. Yemen and Afghanistan are often mentioned in this regard, as are Laos and Cambodia. Allegations they remain. New chemical and biological technologies have produced whole new variations and nobody's bragging about what they may or may not be doing.
A known area of occasional distilled mustard use is in the Iran-Iraq war which began in 1979. When Iraq was pushed back to its starting point by the early 1980s, it is alleged to have begun the use of chemical agents as an effective weapon against Iranian human wave attacks consisting of untrained volunteer levies begging for death and receiving it in spades.
Structure
Distilled mustard has a simple structure, consisting of two ethyl (C2H5) groups bound together around an atom of sulphur. The outer two hydrogen bonds along the central axis have been replaced by chlorine bonds.
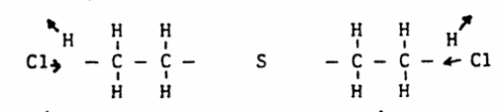
The old Levinstein process consisted of treating ethylene with sulphur chloride.
Pathology
Both distilled mustard and its vapors create an extreme hazard. The greater the absorbed dose of either the greater the damage.
Vapors of this agent will cause temporary blindness and inflammation of the entire respiratory tract. Further heavy vapor exposure will make the blindness permanent and will strip the bronchial tubes of their mucus membrane lining. Any concentration will cause a severe choking effect.
Distilled mustard liquid is corrosive to human tissue both locally and systematic. Local effects include immediate inflammation of the tissues around the eyes and pronounced reddening of exposed skin. If not decontaminated, reddened skin will ulcerate into waterish boils within four to six hours. These blisters, if crudely broken, will reblister. Systemic effects from prolonged exposure may include internal inflammation and blistering (ulceration) of the throat and lungs, resulting in what is termed "dry land drowning," in which the windpipe clogs from bottom to top. Ingestion will cause nausea and vomiting within the same time frame. Absorption into the blood, results in white blood cell destruction. Long term exposures may promote bone marrow destruction and subsequent damage to the immune system. Currently listed by NIOSH/RTECS as being mutagenic in all mammals in solution concentrations above 750 m/liter.
Field behavior
Distilled mustard is among the most commonly listed military casualty agents. It is used to deny terrain and to contaminate equipment and stores.
The delayed effects of distilled mustard necessitate constant chemical survey and monitoring. The human body slowly detoxifies this agent, so prolonged toxic exposures may slowly build up without initial warning symptoms. The vapors are heavier than air and may persist as long as one month in winter weather. They will seek lower elevations in open terrain and substructures, tunnels, and conduits in man-made terrain.
With a flash point of 105°C (221°F) distilled mustard, and particularly its vapors, may explode if exposed to fire or munitions detonations.
Decontamination must be immediate and thorough when this agent is discovered. Exposed personnel should receive immediate personal decontamination and medical examination.
Slightly soluble in cold water, although volatile in steam. Soluble in most organic solvents.
Specific data
median lethal dosage (LCt50)
(Inhalation) 1500 mg-min/m3 or 23cppm/10 molar.
(Skin absorption) 10,000 mg-min/m3 or 34 mg/kg.
median incapacitating dosage (ICt50)
(Eye injury) 200 mg-min/m3.
(Skin absorption) 2000 mg-min/m3.
eye toxicity
Very susceptible to low concentrations (see above). Damage is accumulative to tissue and blood vessels, causing blindness or permanent impairment if untreated.
skin toxicity
Less susceptible to concentrations, but accumulative and irritating. Severe blistering is possible and permanent damage to skin tissues may result even from delayed treatment. HD dissolved in sweat is of great danger to areas of skin like the face, underarms, knees, elbows, and crotch!
rate of action
Delayed. Major pathological symptoms commonly do not appear until at least four hours after exposure. However, due to the accumulative effect of this compound, symptoms may manifest perhaps a week or more after a series of incremental exposures. Effects from ingestion and eye exposure may be more immediate.
protection required
Protective mask/respirator, with full protective overclothing under all conditions.
rate of detoxification
Very low natural detoxification in the body makes the dosages (and effects) accumulative.
decontaminant
Strong bleach solutions and caustic soda (sodium hypochlorite) for personnel and terrain. For the decontamination of buildings and substructures live steam may also be used. Under combat conditions fire may be used as a field expedient decontaminant, mindful of an explosive hazard.
vapor density
5.4 times heavier than air.
vapor pressure
0.072 mm Hg at 20°C (68°F).
liquid density
1.27 g/cc.
persistency
Heavily splashed liquid may last several days in temperate climates. May last up to a month under winter conditions.
volatility
(Solid) 75 mg/m3 at 0°C (32°F), (liquid) 610 mg/m3 at 20°C (68°F), 2680 mg/m3 at 40°C (104°F).
latent heat of vaporization
94 calories per gram
melting point
14.45°C (58.1°F).
boiling point
217°C (422. 6°F) with decomposition.
decomposition point
Begins at 149°C (300°F).
flash point
105°C (221°F).
rate of hydrolysis
Hydrolyses within 17 minutes in distilled water at 25°C (77°F) and two hours in salt water at the same temperature.
hydrolysis products
Hydrochloric acid and thiodiglycol.
stability in storage
Stable in steel or aluminum canisters.
actions on metals or other materials
Little corrosive property when pure. Electrostatic properties in electrical equipment.
molecular weight
159.08
Warning
The above specific data refer to distilled mustard in its traditional form of a pure chemical. Microencapsulation techniques could make this into a highly persistent poison presenting a profoundly greater hazard.
_______________
Notes:
1. During WWI HD was variously known as HS (Britain), das Lost (Germany), and Yprite (France). International codes became standardized by convention after 1919-1920.

Distilled Mustard
HD
Standard NATO agreement (STANAG) code: HD
Chemical name: bis (2-chloroethyl) sulfide Common name: distilled mustard [1] Formula: C1(CH2)2S(CH2)2C1
Family: casualty agent
Type: blister agent (mustard)
An amber brown liquid with an odor similar to that of burning garlic, the odor becoming more pronounced with impurities in the solution. Creates a lowlaying colorless vapor around the splashed liquid.
History
Distilled mustard (HD) was known as early as the late 1880s as a by-product of the dye industry whose toxic effects might be of use in both treating minor tumors and in killing warehouse pests.
Originally investigated by the British and rejected as not being lethal enough, it was first used against them. At 10 PM on the evening of July 12th, 1917 the British 15th and 55th infantry divisions came under an artillery barrage in their positions near Ypres, France. Mixed in with high explosives was a significant quantity of distilled mustard. Other than a stench "like garlic" of "like mustard" (which is how the compound gets its name) there was little nuisance beyond eye irritation like diluted phosgene. Many troops did not bother to put their gas masks on. By the following afternoon the British field hospitals were clogged and on the next evening the first deaths began.
Distilled mustard, the first of the major blister agents, became the chemical compound by which all others have been judged since. According to a British army study, the Foulkes Papers, 16-1/2% of all encountered casualties in the last eighteen months of that war were due to chemicals, mainly distilled mustard. In 1920 about 19,000 British veterans drew permanent disability, mainly from mustard. A 1927 Porton Down study of HD victims found pre-cardiac conditions common, and typical conjunctivitis, laryngitis, bronchitis, and reoccurring skin burns. A Porton Down study of 1929 of 29 severe cases showed fibrosis, respiratory and spinal TB, persistent laryngitis, anemia, conjunctivitis, aphonia, and pulmonary fibrosis. By 1930, 80% of known survivors suffered chronic bronchitis.
After the first world war, limited production continued in Great Britain and France. Production began in Italy and the Soviet Union by 1925, and in Japan in 1928.
HD was allegedly used against the Afghans by the British in 1919. It was known to be used against Moroccans by the French and Spanish in 1925, against Ethiopians by the Italians after 1935, and by the Japanese against China after 1934 and ending by 1944.
The late 1920s through the 1930s were a time a chemical warfare experimentation as the older agents were adapted to fit new methods and tactics of war. New munitions, especially those for aerial sprays and bombs, were developed. It was assumed that chemicals would be used in the the "next war" to the same extent they had been used in 1915-1918. As German rearmament beginning in 1934 started a general trend, chemical munitions plants were upgraded.
Great Britain built new facilities such as those at Sutton Oak-St Helens, Lancashire. France opened a HD and CG plant at Clancy. The USSR built new HD-L-CG plants at Brandyuzhsky, Kuibyshev, and Karaganda. Germany built plants at Munster, Wunsdorf, and List. The U.S. reluctantly reopened Pine Bluff Arsenal (in production by 1942) and built new facilities such as Rocky Mountain Arsenal (near Denver) and opened the Dugway, Utah, Proving Grounds for the express purpose of testing chemical agents.
Casualty agent use in the second world war, outside of China through 1943-1944, was virtually non-existent. The only known incident in the west occurred in Bari harbor on the Italian Adriatic.
An American merchantman, the S.S. John Harvey, carried the bulk of U.S. chemical munitions for the Mediterranean area, in the form of 2000 M47A1 aerial bombs, containing a total of about 100 tons of HD. The S.S. John Harvey's cargo was known only to SHAFE in London and a few junior Army Ordinance officers on board. On 7:30 PM, July 2nd, 1943, the John Harvey was sunk in a German air raid. Her cargo of HD had all been made by the quick-but-dirty Levinstein process. Thus it had hydrogen and ethane gas impurities in the HD mix. The John Harvey blew up and flooded the harbor. By the following morning it was Ypres all over again, except nobody knew why the massive blistering was happening, especially among sailors soaked in oil and some smelly stuff from the harbor waters. About 630 serious military cases occurred and over a thousand Italian civilians died within a few days.
Allegations of the use of distilled mustard surface from time to time. Yemen and Afghanistan are often mentioned in this regard, as are Laos and Cambodia. Allegations they remain. New chemical and biological technologies have produced whole new variations and nobody's bragging about what they may or may not be doing.
A known area of occasional distilled mustard use is in the Iran-Iraq war which began in 1979. When Iraq was pushed back to its starting point by the early 1980s, it is alleged to have begun the use of chemical agents as an effective weapon against Iranian human wave attacks consisting of untrained volunteer levies begging for death and receiving it in spades.
Structure
Distilled mustard has a simple structure, consisting of two ethyl (C2H5) groups bound together around an atom of sulphur. The outer two hydrogen bonds along the central axis have been replaced by chlorine bonds.
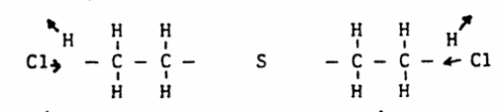
The old Levinstein process consisted of treating ethylene with sulphur chloride.
Pathology
Both distilled mustard and its vapors create an extreme hazard. The greater the absorbed dose of either the greater the damage.
Vapors of this agent will cause temporary blindness and inflammation of the entire respiratory tract. Further heavy vapor exposure will make the blindness permanent and will strip the bronchial tubes of their mucus membrane lining. Any concentration will cause a severe choking effect.
Distilled mustard liquid is corrosive to human tissue both locally and systematic. Local effects include immediate inflammation of the tissues around the eyes and pronounced reddening of exposed skin. If not decontaminated, reddened skin will ulcerate into waterish boils within four to six hours. These blisters, if crudely broken, will reblister. Systemic effects from prolonged exposure may include internal inflammation and blistering (ulceration) of the throat and lungs, resulting in what is termed "dry land drowning," in which the windpipe clogs from bottom to top. Ingestion will cause nausea and vomiting within the same time frame. Absorption into the blood, results in white blood cell destruction. Long term exposures may promote bone marrow destruction and subsequent damage to the immune system. Currently listed by NIOSH/RTECS as being mutagenic in all mammals in solution concentrations above 750 m/liter.
Field behavior
Distilled mustard is among the most commonly listed military casualty agents. It is used to deny terrain and to contaminate equipment and stores.
The delayed effects of distilled mustard necessitate constant chemical survey and monitoring. The human body slowly detoxifies this agent, so prolonged toxic exposures may slowly build up without initial warning symptoms. The vapors are heavier than air and may persist as long as one month in winter weather. They will seek lower elevations in open terrain and substructures, tunnels, and conduits in man-made terrain.
With a flash point of 105°C (221°F) distilled mustard, and particularly its vapors, may explode if exposed to fire or munitions detonations.
Decontamination must be immediate and thorough when this agent is discovered. Exposed personnel should receive immediate personal decontamination and medical examination.
Slightly soluble in cold water, although volatile in steam. Soluble in most organic solvents.
Specific data
median lethal dosage (LCt50)
(Inhalation) 1500 mg-min/m3 or 23cppm/10 molar.
(Skin absorption) 10,000 mg-min/m3 or 34 mg/kg.
median incapacitating dosage (ICt50)
(Eye injury) 200 mg-min/m3.
(Skin absorption) 2000 mg-min/m3.
eye toxicity
Very susceptible to low concentrations (see above). Damage is accumulative to tissue and blood vessels, causing blindness or permanent impairment if untreated.
skin toxicity
Less susceptible to concentrations, but accumulative and irritating. Severe blistering is possible and permanent damage to skin tissues may result even from delayed treatment. HD dissolved in sweat is of great danger to areas of skin like the face, underarms, knees, elbows, and crotch!
rate of action
Delayed. Major pathological symptoms commonly do not appear until at least four hours after exposure. However, due to the accumulative effect of this compound, symptoms may manifest perhaps a week or more after a series of incremental exposures. Effects from ingestion and eye exposure may be more immediate.
protection required
Protective mask/respirator, with full protective overclothing under all conditions.
rate of detoxification
Very low natural detoxification in the body makes the dosages (and effects) accumulative.
decontaminant
Strong bleach solutions and caustic soda (sodium hypochlorite) for personnel and terrain. For the decontamination of buildings and substructures live steam may also be used. Under combat conditions fire may be used as a field expedient decontaminant, mindful of an explosive hazard.
vapor density
5.4 times heavier than air.
vapor pressure
0.072 mm Hg at 20°C (68°F).
liquid density
1.27 g/cc.
persistency
Heavily splashed liquid may last several days in temperate climates. May last up to a month under winter conditions.
volatility
(Solid) 75 mg/m3 at 0°C (32°F), (liquid) 610 mg/m3 at 20°C (68°F), 2680 mg/m3 at 40°C (104°F).
latent heat of vaporization
94 calories per gram
melting point
14.45°C (58.1°F).
boiling point
217°C (422. 6°F) with decomposition.
decomposition point
Begins at 149°C (300°F).
flash point
105°C (221°F).
rate of hydrolysis
Hydrolyses within 17 minutes in distilled water at 25°C (77°F) and two hours in salt water at the same temperature.
hydrolysis products
Hydrochloric acid and thiodiglycol.
stability in storage
Stable in steel or aluminum canisters.
actions on metals or other materials
Little corrosive property when pure. Electrostatic properties in electrical equipment.
molecular weight
159.08
Warning
The above specific data refer to distilled mustard in its traditional form of a pure chemical. Microencapsulation techniques could make this into a highly persistent poison presenting a profoundly greater hazard.
_______________
Notes:
1. During WWI HD was variously known as HS (Britain), das Lost (Germany), and Yprite (France). International codes became standardized by convention after 1919-1920.
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-10
Chemical Agent Monitor (CAM) in use

Chemical Agent Monitor (CAM) in use
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-11
DEPARTMENT OF THE ARMY
Headquarters, Task Force VICTORY (Fwd)
Camp Doha, Kuwait
APO 09889-0003
AETSBGC-V
7 August 1991
MEMORANDUM FOR Commander, 11th ACR, ATTN: RS3
Subject: Tasking Number 91-047
1. You are tasked to provide the following support: Two FOX NBC Reconnaissance Vehicles in support of Kuwaiti MOD and British EOD.
2. Personnel: Personnel to operate 2 FOX NBC Reconnaissance Vehicles.
3. Equipment required: 2 FOX Reconnaissance Vehicles.
4. Specific instructions:
a. Initially FOX Vehicles will be used to provide NBC reconnaissance/detection.
b. On order be prepared to provide two FOX NBC Reconnaissance Vehicles for escort/monitoring of EOD operations.
c. Standard NBC SOPs will be followed to ensure safety of FOX vehicle crews.
d. Direct coordination with Major Jon Watkinson, British Royal Engineers, Commander 21st EOD Group, Beteal Camp Messlack, grid 181376, 539-4505 (Comm) is authorized.
e. Report status to TF Victory POC LTC Killgore, 5056 (AT&T).
5. The attached report from the British Army is provided for your information.
JOSEPH W. MILLER
LTC, GS
ACofS; G-3
CF:
Chief of Staff, TF Victory (Fwd)
DEPARTMENT OF THE ARMY
Headquarters, Task Force VICTORY (Fwd)
Camp Doha, Kuwait
APO 09889-0003
AETSBGC-V
7 August 1991
MEMORANDUM FOR Commander, 11th ACR, ATTN: RS3
Subject: Tasking Number 91-047
1. You are tasked to provide the following support: Two FOX NBC Reconnaissance Vehicles in support of Kuwaiti MOD and British EOD.
2. Personnel: Personnel to operate 2 FOX NBC Reconnaissance Vehicles.
3. Equipment required: 2 FOX Reconnaissance Vehicles.
4. Specific instructions:
a. Initially FOX Vehicles will be used to provide NBC reconnaissance/detection.
b. On order be prepared to provide two FOX NBC Reconnaissance Vehicles for escort/monitoring of EOD operations.
c. Standard NBC SOPs will be followed to ensure safety of FOX vehicle crews.
d. Direct coordination with Major Jon Watkinson, British Royal Engineers, Commander 21st EOD Group, Beteal Camp Messlack, grid 181376, 539-4505 (Comm) is authorized.
e. Report status to TF Victory POC LTC Killgore, 5056 (AT&T).
5. The attached report from the British Army is provided for your information.
JOSEPH W. MILLER
LTC, GS
ACofS; G-3
CF:
Chief of Staff, TF Victory (Fwd)
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-12
ASSISTANT TO THE SECRETARY OF DEFENSE
30500 DEFENSE PENTAGON
WASHINGTON. DC 2030-3050
The Honorable Donald W. Riegle. Jr.
Chairman
Committee on Banking, Housing, and Urban Affairs
United States Senate
Washington. D.C. 20510-6075
Dear Senator Riegle:
Secretary Perry has designated me the point of contact for all Operation Desert Storm chemical and biological weapons matters. It is in this capacity I am providing an interim response to your July 5, 1994 letter to Major General Jerry Harrison concerning the alleged discovery of suspected Iraqi chemical agents in 1991.
CPT Michael F. Johnson's statement is at Enclosure 1. Included in CPT Johnson's statement are the initial British report and photocopies of photographs. CPT Johnson was not aware that samples had been collected from the site for analysis at Porton Down, or that a special United Nations chemical weapons evaluation team had responded to the site.
This incident was discussed with COL Macel, Central Army Command J4/7, in April 1994 (Enclosure 2). COL Macel was the Chief of Security Assistance and senior U.S. Defense representative in Kuwait following the cease fire. COL Macel was present at the site during efforts to identify the liquid. He knew that a United Nations chemical warfare specialist team was flown in from Bahrain to assess the liquid, and that samples of the liquid were flown to Great Britain for further analysis.
A statement from Dr. Graham S. Pearson, Director General, Chemical & Biological Defence Establishment, United Kingdom Ministry of Defence is expected to be released by the British Government early this week. Dr. Pearson telephonically related that a memorandum from Porton Down to the Ministry of Defence dated August 23, 1991 stated that the brown fumes emitting from the tank, destruction of the protective suit material, and the skin blister suggested that the liquid was most likely fuming nitric acid, a highly corrosive oxidizing acid used as a rocket propellant, and not a chemical warfare agent. Iraq used red fuming nitric acid as a rocket propellant during the war.
Samples of the suspected agent and overgarment material were delivered to Porton Downs for analysis on 13 September 1991. Analytical results showed that there was a high concentration of nitrate in the sample and a PH that was extremely acidic. The scientific analysis determined that the contents of the tank were nitric acid and there was no evidence of any CW agent being present.
The Fox reconnaissance vehicle tapes were analyzed by the Program Manager NBC Defense Systems, (PM NBC) Fox Reconnaissance Vehicle Division. Analysis of the molecular ions showed that the ions matched in three of four categories for H agent, and were not in the correct proportions for H or phosgene oxime. When the PM NBC scientists beard of the British identifying nitric acid they compared the Fox tapes to the spectrometry of nitric acid. The spectrum matched nitric acid ions in all four categories, and were in the correct proportions.
All personnel at the scene of the suspected chemical agent performed their duties in an exemplary manner. Proper planning and coordination was made between U.S., British and U.N. forces; all field equipment was used properly; all technical resources were employed; and following proper NATO procedures, samples were taken and transported for laboratory analysis.
Indicators were present that could have caused soldiers on the scene to identify the liquid as an industrial chemical were simply overlooked because of the positive readings received on the chemical detection test equipment. The blister injury suffered by the British soldier was an indicator that the liquid was not H mustard or phosgene oxime. The soldier was in extreme pain and a blister formed within one minute of agent contact. H mustard agents are insidious, pain and symptoms are delayed for a minimum of one hour and usually 4-12 hours. Phosgene oxime blanches the skin within 30 seconds but does not cause blisters. See Enclosure 3 for more explanation. Red fuming nitric acid causes extreme pain and blisters upon contact with the skin. In a telephonic conversation with CPT Johnson on July 15, 1994 he stated that the chemical protective gloves worn by soldiers began disintegrating upon contact with the liquid. U.S. chemical protective gloves are designed to withstand contact with liquid agent and would be unaffected. Nitric acid would cause immediate disintegration.
Fuming nitric acid is identifiable by its tell-tale brown fuming smoke.
We anticipate receiving additional information next week that will completely answer your inquiry. PM NBC Defense is searching its files for the memorandum it prepared on the analysis of the tapes. As a cross- check, CPT Johnson will provide the vehicle identification numbers so historical tapes from the Fox vehicles can be compared against the tapes analyzed by PM NBC Defense. The British Chemical and Biological Defense Establishment recommended that the nitric acid be disposed of by selling it to a local chemical industry or pay to have it neutralized under controlled conditions. Attempts will be made to verify its method of disposal.
Research and correct interpretation of incidents of this nature is a sometimes difficult and tedious task. Our Chemical Section is always available to analyze and explain the technical and operational aspects of reported chemical warfare related incidents. We appreciate the opportunity to provide you with this information.
THEODORE M. PROCIV
Deputy for Chemical/Biological Matters
Enclosures
[Handwritten Note: Sorry about the quality of the pictures ... we only have copies, no originals. TP]
ASSISTANT TO THE SECRETARY OF DEFENSE
30500 DEFENSE PENTAGON
WASHINGTON. DC 2030-3050
The Honorable Donald W. Riegle. Jr.
Chairman
Committee on Banking, Housing, and Urban Affairs
United States Senate
Washington. D.C. 20510-6075
Dear Senator Riegle:
Secretary Perry has designated me the point of contact for all Operation Desert Storm chemical and biological weapons matters. It is in this capacity I am providing an interim response to your July 5, 1994 letter to Major General Jerry Harrison concerning the alleged discovery of suspected Iraqi chemical agents in 1991.
CPT Michael F. Johnson's statement is at Enclosure 1. Included in CPT Johnson's statement are the initial British report and photocopies of photographs. CPT Johnson was not aware that samples had been collected from the site for analysis at Porton Down, or that a special United Nations chemical weapons evaluation team had responded to the site.
This incident was discussed with COL Macel, Central Army Command J4/7, in April 1994 (Enclosure 2). COL Macel was the Chief of Security Assistance and senior U.S. Defense representative in Kuwait following the cease fire. COL Macel was present at the site during efforts to identify the liquid. He knew that a United Nations chemical warfare specialist team was flown in from Bahrain to assess the liquid, and that samples of the liquid were flown to Great Britain for further analysis.
A statement from Dr. Graham S. Pearson, Director General, Chemical & Biological Defence Establishment, United Kingdom Ministry of Defence is expected to be released by the British Government early this week. Dr. Pearson telephonically related that a memorandum from Porton Down to the Ministry of Defence dated August 23, 1991 stated that the brown fumes emitting from the tank, destruction of the protective suit material, and the skin blister suggested that the liquid was most likely fuming nitric acid, a highly corrosive oxidizing acid used as a rocket propellant, and not a chemical warfare agent. Iraq used red fuming nitric acid as a rocket propellant during the war.
Samples of the suspected agent and overgarment material were delivered to Porton Downs for analysis on 13 September 1991. Analytical results showed that there was a high concentration of nitrate in the sample and a PH that was extremely acidic. The scientific analysis determined that the contents of the tank were nitric acid and there was no evidence of any CW agent being present.
The Fox reconnaissance vehicle tapes were analyzed by the Program Manager NBC Defense Systems, (PM NBC) Fox Reconnaissance Vehicle Division. Analysis of the molecular ions showed that the ions matched in three of four categories for H agent, and were not in the correct proportions for H or phosgene oxime. When the PM NBC scientists beard of the British identifying nitric acid they compared the Fox tapes to the spectrometry of nitric acid. The spectrum matched nitric acid ions in all four categories, and were in the correct proportions.
All personnel at the scene of the suspected chemical agent performed their duties in an exemplary manner. Proper planning and coordination was made between U.S., British and U.N. forces; all field equipment was used properly; all technical resources were employed; and following proper NATO procedures, samples were taken and transported for laboratory analysis.
Indicators were present that could have caused soldiers on the scene to identify the liquid as an industrial chemical were simply overlooked because of the positive readings received on the chemical detection test equipment. The blister injury suffered by the British soldier was an indicator that the liquid was not H mustard or phosgene oxime. The soldier was in extreme pain and a blister formed within one minute of agent contact. H mustard agents are insidious, pain and symptoms are delayed for a minimum of one hour and usually 4-12 hours. Phosgene oxime blanches the skin within 30 seconds but does not cause blisters. See Enclosure 3 for more explanation. Red fuming nitric acid causes extreme pain and blisters upon contact with the skin. In a telephonic conversation with CPT Johnson on July 15, 1994 he stated that the chemical protective gloves worn by soldiers began disintegrating upon contact with the liquid. U.S. chemical protective gloves are designed to withstand contact with liquid agent and would be unaffected. Nitric acid would cause immediate disintegration.
Fuming nitric acid is identifiable by its tell-tale brown fuming smoke.
We anticipate receiving additional information next week that will completely answer your inquiry. PM NBC Defense is searching its files for the memorandum it prepared on the analysis of the tapes. As a cross- check, CPT Johnson will provide the vehicle identification numbers so historical tapes from the Fox vehicles can be compared against the tapes analyzed by PM NBC Defense. The British Chemical and Biological Defense Establishment recommended that the nitric acid be disposed of by selling it to a local chemical industry or pay to have it neutralized under controlled conditions. Attempts will be made to verify its method of disposal.
Research and correct interpretation of incidents of this nature is a sometimes difficult and tedious task. Our Chemical Section is always available to analyze and explain the technical and operational aspects of reported chemical warfare related incidents. We appreciate the opportunity to provide you with this information.
THEODORE M. PROCIV
Deputy for Chemical/Biological Matters
Enclosures
[Handwritten Note: Sorry about the quality of the pictures ... we only have copies, no originals. TP]
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-13
United States Senate
COMMITTEE ON BANKING, HOUSING, AND URBAN AFFAIRS
WASHINGTON, DC 20150-6075
August 1, 1994
Mr. Stephen Stein
National Institutes of Standards and Technology
Spectrometry Standards
NIST A260/222
Gaithersburg, Maryland 20899
Dear Mr. Stein:
Recently, the Department of Defense responded to a congressional request for explanation of a U.S. report prepared after the Persian Gulf War that details the identification of bis (2-chloroethyl) sulfide (C1(CH2)2S(CH2)2C1], carbonyl chloride [COCl2], and dichloroform oxime [CCl2NOH]. The official Department of Defense explanation was that the substance was actually fuming red nitric acid. The identifications for bis (2-chloroethyl) sulfide, sulfur mustard, were made by an array of military chemical identification equipment as disclosed in the attached reports. The final eight identifications (4-four peak identifications and 4 full spectra identifications) were made by two different Bruker Instruments MM1 mass spectrometers. According to the manufacturer, the following four principal peaks were programmed into the computer's identification algorithm for recognition of these substances:
For identification of bis (2-cholroethyl) sulfide [Cl(CH2)2S(CH2)2Cl]; common name: sulfur mustard (HD) Molecular Weight: 159.08
158.0 (rel. intensity 20.1%), 109.0 (rel. intensity 100%).
111.0 (rel. intensity 41.6%), and 160.0 (rel. intensity 12.9%).
For identification of carbonyl chloride (COCl2)]; common name: phosgene (CG) Molecular Weight: 98.92
65.0 (rel. intensity 38.8%), 63.0 (rel. intensity 100%),
98.0 (rel. intensity 11.2%), and 109.0 (rel. intensity 0.0%). For identification of dichloroform oxime [CCl2NOH]; common name: phosgene oxime (CX) Molecular Weight: 113.92
113.0 (rel. intensity 55.9%), 78.0 (rel. intensity 100%),
115.0 (rel. intensity 38.8%), and 77.0 (rel. intensity 55.9%).
The mass of fuming red nitric acid, HNO3, is 63.01 and its most likely breakdown components appear to be nitrogen oxides and hydrogen of even lesser molecular weights. The sample of the questioned liquid was drawn directly from the container using a standard plastic medical syringe with a standard plastic medical i.v. tubing, deposited into a metallic dish, and transported directly to the ground probe of the MM1. This ground probe has a chrome nickel cap, and there was no damage to either the metal dish or the chrome nickel cap on the ground probe.
The MM1 identified sulfur mustard (HD) with a 6.4 confirmation reading. This, according to the manufacturer, indicates highly concentrated HD agent.
Given the foregoing:
Q.1. What are the principal mass peaks for fuming red nitric acid (HNO3)?
Q.2. Is it possible that a mass spectrometer could match 3 of 4 peaks or 4 of 4 peaks for HD all of which appear above the total mass of nitric acid and still match 4 of 4 for fuming red nitric acid?
Q.3. Is the reverse possible, that is, could the mass peaks for HNO3 be confused with those for HD? Under what circumstances might this be possible? Is it likely?
Q.4. The commander of the unit said that tests were run using both the 4 principle mass peaks and full spectrum analysis on the questioned substances. The tests were run twice each by two FOX vehicles using the MM1. These MM1s were checked for calibration both before and after the tests were run and with no problems noted. Further each time the testing was conducted identical substances were repeatedly identified, chemical mustard agent, phosgene, and phosgene oxime. How likely is it that under these circumstances the computer algorithm identified these chemicals instead of fuming red nitric acid?
Q.5. Bruker Instruments has contacted Thyssen Henschel regarding conducting a field trial with fuming red nitric acid on an operational FOX vehicle and said they responded with some concern that it might result in damage to the GC/MS. Could fuming red nitric acid (conc. >90%) damage a mass spectrometer? Be specific.
Q.6. Does NIST maintain a spectrum standard for nitric acid. If not, why?
Q.7. If a metallic container were used to store nitric acid, what kind of metal container would be required? If two open bullet holes were in the container for over 5 months, that is, if there were a continuous source of oxygen to the fuming red nitric acid, would it evaporate?
Q.8. If a metal tray were used to transport red fuming nitric acid from this container to a mass spectrometer, would it react with the metal? What metals would it not react with?
Any assistance either you or any other qualified scientists at the National Institutes of Standards and Technology might provide in answering these questions would be appreciated. Please feel free to contact me directly at (202)-224-1563, if you have any questions regarding this request.
Sincerely,
James J. Tuite
Professional Staff
Attachments
United States Senate
COMMITTEE ON BANKING, HOUSING, AND URBAN AFFAIRS
WASHINGTON, DC 20150-6075
August 1, 1994
Mr. Stephen Stein
National Institutes of Standards and Technology
Spectrometry Standards
NIST A260/222
Gaithersburg, Maryland 20899
Dear Mr. Stein:
Recently, the Department of Defense responded to a congressional request for explanation of a U.S. report prepared after the Persian Gulf War that details the identification of bis (2-chloroethyl) sulfide (C1(CH2)2S(CH2)2C1], carbonyl chloride [COCl2], and dichloroform oxime [CCl2NOH]. The official Department of Defense explanation was that the substance was actually fuming red nitric acid. The identifications for bis (2-chloroethyl) sulfide, sulfur mustard, were made by an array of military chemical identification equipment as disclosed in the attached reports. The final eight identifications (4-four peak identifications and 4 full spectra identifications) were made by two different Bruker Instruments MM1 mass spectrometers. According to the manufacturer, the following four principal peaks were programmed into the computer's identification algorithm for recognition of these substances:
For identification of bis (2-cholroethyl) sulfide [Cl(CH2)2S(CH2)2Cl]; common name: sulfur mustard (HD) Molecular Weight: 159.08
158.0 (rel. intensity 20.1%), 109.0 (rel. intensity 100%).
111.0 (rel. intensity 41.6%), and 160.0 (rel. intensity 12.9%).
For identification of carbonyl chloride (COCl2)]; common name: phosgene (CG) Molecular Weight: 98.92
65.0 (rel. intensity 38.8%), 63.0 (rel. intensity 100%),
98.0 (rel. intensity 11.2%), and 109.0 (rel. intensity 0.0%). For identification of dichloroform oxime [CCl2NOH]; common name: phosgene oxime (CX) Molecular Weight: 113.92
113.0 (rel. intensity 55.9%), 78.0 (rel. intensity 100%),
115.0 (rel. intensity 38.8%), and 77.0 (rel. intensity 55.9%).
The mass of fuming red nitric acid, HNO3, is 63.01 and its most likely breakdown components appear to be nitrogen oxides and hydrogen of even lesser molecular weights. The sample of the questioned liquid was drawn directly from the container using a standard plastic medical syringe with a standard plastic medical i.v. tubing, deposited into a metallic dish, and transported directly to the ground probe of the MM1. This ground probe has a chrome nickel cap, and there was no damage to either the metal dish or the chrome nickel cap on the ground probe.
The MM1 identified sulfur mustard (HD) with a 6.4 confirmation reading. This, according to the manufacturer, indicates highly concentrated HD agent.
Given the foregoing:
Q.1. What are the principal mass peaks for fuming red nitric acid (HNO3)?
Q.2. Is it possible that a mass spectrometer could match 3 of 4 peaks or 4 of 4 peaks for HD all of which appear above the total mass of nitric acid and still match 4 of 4 for fuming red nitric acid?
Q.3. Is the reverse possible, that is, could the mass peaks for HNO3 be confused with those for HD? Under what circumstances might this be possible? Is it likely?
Q.4. The commander of the unit said that tests were run using both the 4 principle mass peaks and full spectrum analysis on the questioned substances. The tests were run twice each by two FOX vehicles using the MM1. These MM1s were checked for calibration both before and after the tests were run and with no problems noted. Further each time the testing was conducted identical substances were repeatedly identified, chemical mustard agent, phosgene, and phosgene oxime. How likely is it that under these circumstances the computer algorithm identified these chemicals instead of fuming red nitric acid?
Q.5. Bruker Instruments has contacted Thyssen Henschel regarding conducting a field trial with fuming red nitric acid on an operational FOX vehicle and said they responded with some concern that it might result in damage to the GC/MS. Could fuming red nitric acid (conc. >90%) damage a mass spectrometer? Be specific.
Q.6. Does NIST maintain a spectrum standard for nitric acid. If not, why?
Q.7. If a metallic container were used to store nitric acid, what kind of metal container would be required? If two open bullet holes were in the container for over 5 months, that is, if there were a continuous source of oxygen to the fuming red nitric acid, would it evaporate?
Q.8. If a metal tray were used to transport red fuming nitric acid from this container to a mass spectrometer, would it react with the metal? What metals would it not react with?
Any assistance either you or any other qualified scientists at the National Institutes of Standards and Technology might provide in answering these questions would be appreciated. Please feel free to contact me directly at (202)-224-1563, if you have any questions regarding this request.
Sincerely,
James J. Tuite
Professional Staff
Attachments
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Re: United States Dual-Use Exports to Iraq and Their Impact
APPENDIX B-14
NIST
UNITED STATES DEPARTMENT OF COMMERCE
National Institute of Standards and Technology
Gaithersburg, Maryland 20899-0001
SEP - 6 1994
Bldg. 222/Rm. A260
Mr. James J. Tuite
Committee on Banking, Housing and Urban Affairs
United Sates Senate
Washington, DC 20510-6075
Dear Mr. Tuite:
In this letter I will answer, as fully as I can, the questions posed in your letter of August 1, 1994 concerning the possible detection of chemical agents by Bruker Instruments MM1 mass spectrometers after the Persian Gulf War. In preparing this response, I have reviewed the documents that you provided, consulted with several NIST colleagues and conducted a literature search. Responses to each of your eight questions are presented below, followed by a discussion of the difficulties expected in the detection of nitric acid by mass spectrometry.
Q.1. What are the principal mass peaks for fuming red nitric acid (HNO3)?
A.1. Fuming red nitric acid is a mixture of two compounds, nitric acid (HNO3) and nitrogen dioxide (NO2). For nitrogen dioxide, the largest peak (100%) appears at 30 u, the next largest is the molecular ion at 46 u (37%). Smaller peaks are present at 16 u (22%) and 14 u (10%). 1 was unable to locate a reference spectrum for nitric acid. However, because of the instability of its molecular ion, I do not expect a significant molecular ion peak at 63 u. Instead, I would expect the same peaks as nitrogen dioxide in approximately the same magnitudes, possibly accompanied by additional small peaks at 62 u (NO3) and 17 u (OH*).
Q.2. Is it possible that a mass spectrometer could match 3 of 4 peaks or 4 of 4 peaks for HD all of which appear above the total mass of nitric acid and still match 4 of 4 for fuming red nitric acid?
A.2. HD has no major peaks in common with those expected to arise directly from fuming nitric acid. It is highly unlikely that a properly functioning mass spectrometer would produce any of the major peaks characteristic of HNO3 or NO2 from HD.
Q.3. Is the reverse possible, that is, could the mass peaks for HNO3, be confused with those for HD? Under what circumstances might this be possible? Is it likely?
A.3. If fuming red nitric acid did not decompose prior to detection (ionization), there would be no possibility of mistaking it for HD. Under proper operating conditions, it could not generate significant peaks above the molecular ion. If, however, the nitric acid underwent chemical reaction prior to detection, its decomposition products might fortuitously give rise to peaks in the mass spectrum of HD. It is highly unlikely, however, that these decomposition products would generate a spectrum that resembles that of HD.
Q.4. The commander of the unit said that tests were run using both the 4 principle mass peaks and full spectrum analysis on the questioned substances. The tests were run twice each by two FOX vehicles using MM1. These MM1s were checked for calibration both before and after the tests were run and with no problems noted. Further each time the testing was conducted identical substances were repeatedly identified, chemical mustard agent, phosgene, and phosgene oxime. How likely is it that under these circumstances the computer algorithm identified these chemicals instead of fuming ted nitric acid?
A.4. If fuming red nitric acid did not react prior to detection, there is no likelihood that either the 4 peak analyses or full spectrum analyses would lead to the false identification of HD. However, if nitric acid did react, the reaction products might generate a large number of peaks. Some of these might, fortuitously, be those characteristic of HD or other chemical agents and therefore might produce a false positive 4-peak identification of HD. A robust, full-spectrum matching algorithm, however, would not be expected to falsely identify HD. However, being unfamiliar with the algorithms used by the MM1 mass spectrometer, I cannot further evaluate this possibility.
Q.5. Bruker Instruments has contacted Thyssen Hanschel regarding conducting a field trial with fuming red nitric acid on an operational FOX vehicle and said they responded with some concern that it might result in damage to the GC/MS. Could fuming red notric acid (conc. >90%) damage a mass spectrometer? Be specific.
A. 5. Red fuming nitric acid could damage a GC/MS, especially one not designed to handle highly reactive materials. Damage could occur in any of the three regions of the GC, the inlet, capillary column or outlet, depending on the materials used for construction or deposited during use. Organic substances, certain metal surfaces or other materials may vigorously react with nitric acid or even nitrogen dioxide. In the mass spectrometer section, reaction at the silicone membrane or even in the high vacuum chamber is also possible.
Q.6. Does NIST maintain a spectrum standard for nitric acid? If not, why?
A.6. We have no reference spectrum for nitric acid in our collection, nor have we been able to find one in other collections or in a cursory literature survey. This absence is not as surprising as one might first imagine for such a common compound. Owing to the high reactivity of nitric acid, the reliable determination of its mass spectrum would probably require special instrumentation and substantial effort. Since mass spectrometry is not a practical method for the identification of nitric acid, there has been little motivation for making this determination.
Q.7. If a metallic container were used to store nitric acid, what kind of metal container would be required? If two open bullet holes were in the container for over 5 months, that is, if there were a continuous source of oxygen to the fuming red nitric acid, would it evaporate?
A.7. Large quantities of red fuming nitric acid are normally shipped and stored in aluminum or stainless steel containers. The volatility of this substance depends on the relative amounts of nitric acid and nitrogen dioxide present, with the normal boiling point varying from 25 C for a 45/55 wt%/wt% NO2/HNO3 mixture to 86 C for pure nitric acid. Once a sufficient amount of nitrogen dioxide has evaporated to raise the boiling point above ambient, further evaporation through holes in a large container would be slow.
Q.8. If a metal tray were used to transport red fuming nitric acid from this container to a mass spectrometer, would it react with the metal? What metals would it not react with?
A.8. As mentioned above, fuming red nitric acid is not reactive towards passivated aluminum or stainless steel (or other chromium-steel alloys). Passivation of these untreated surfaces occurs through a mild chemical reaction.
One issue, that of the detection of nitric acid by mass spectrometry, deserves further comment. Unless a mass spectrometer is specifically designed to detect highly reactive gases, it is not clear that a good quality spectrum nitric acid would be obtained. It is even less likely that nitric acid would pass intact through a GC and then through the silicone membrane inlet of the Bruker instrument to the mass spectrometer. Therefore, the introduction of nitric acid into a GC/MS may produce unpredictable results. Without examining the observed spectra and spectral comparison algorithms, I cannot evaluate the plausibility of the claimed false positive identifications.
Upon request, I can provide further details concerning any the above responses. I will also be glad to provide further technical assistance on these matters.
Sincerely,
Stephen E. Stein, Ph.D.
Director. NIST Mass Spectrometry Data Center
Chemical Science and Technology Laboratory
NIST
UNITED STATES DEPARTMENT OF COMMERCE
National Institute of Standards and Technology
Gaithersburg, Maryland 20899-0001
SEP - 6 1994
Bldg. 222/Rm. A260
Mr. James J. Tuite
Committee on Banking, Housing and Urban Affairs
United Sates Senate
Washington, DC 20510-6075
Dear Mr. Tuite:
In this letter I will answer, as fully as I can, the questions posed in your letter of August 1, 1994 concerning the possible detection of chemical agents by Bruker Instruments MM1 mass spectrometers after the Persian Gulf War. In preparing this response, I have reviewed the documents that you provided, consulted with several NIST colleagues and conducted a literature search. Responses to each of your eight questions are presented below, followed by a discussion of the difficulties expected in the detection of nitric acid by mass spectrometry.
Q.1. What are the principal mass peaks for fuming red nitric acid (HNO3)?
A.1. Fuming red nitric acid is a mixture of two compounds, nitric acid (HNO3) and nitrogen dioxide (NO2). For nitrogen dioxide, the largest peak (100%) appears at 30 u, the next largest is the molecular ion at 46 u (37%). Smaller peaks are present at 16 u (22%) and 14 u (10%). 1 was unable to locate a reference spectrum for nitric acid. However, because of the instability of its molecular ion, I do not expect a significant molecular ion peak at 63 u. Instead, I would expect the same peaks as nitrogen dioxide in approximately the same magnitudes, possibly accompanied by additional small peaks at 62 u (NO3) and 17 u (OH*).
Q.2. Is it possible that a mass spectrometer could match 3 of 4 peaks or 4 of 4 peaks for HD all of which appear above the total mass of nitric acid and still match 4 of 4 for fuming red nitric acid?
A.2. HD has no major peaks in common with those expected to arise directly from fuming nitric acid. It is highly unlikely that a properly functioning mass spectrometer would produce any of the major peaks characteristic of HNO3 or NO2 from HD.
Q.3. Is the reverse possible, that is, could the mass peaks for HNO3, be confused with those for HD? Under what circumstances might this be possible? Is it likely?
A.3. If fuming red nitric acid did not decompose prior to detection (ionization), there would be no possibility of mistaking it for HD. Under proper operating conditions, it could not generate significant peaks above the molecular ion. If, however, the nitric acid underwent chemical reaction prior to detection, its decomposition products might fortuitously give rise to peaks in the mass spectrum of HD. It is highly unlikely, however, that these decomposition products would generate a spectrum that resembles that of HD.
Q.4. The commander of the unit said that tests were run using both the 4 principle mass peaks and full spectrum analysis on the questioned substances. The tests were run twice each by two FOX vehicles using MM1. These MM1s were checked for calibration both before and after the tests were run and with no problems noted. Further each time the testing was conducted identical substances were repeatedly identified, chemical mustard agent, phosgene, and phosgene oxime. How likely is it that under these circumstances the computer algorithm identified these chemicals instead of fuming ted nitric acid?
A.4. If fuming red nitric acid did not react prior to detection, there is no likelihood that either the 4 peak analyses or full spectrum analyses would lead to the false identification of HD. However, if nitric acid did react, the reaction products might generate a large number of peaks. Some of these might, fortuitously, be those characteristic of HD or other chemical agents and therefore might produce a false positive 4-peak identification of HD. A robust, full-spectrum matching algorithm, however, would not be expected to falsely identify HD. However, being unfamiliar with the algorithms used by the MM1 mass spectrometer, I cannot further evaluate this possibility.
Q.5. Bruker Instruments has contacted Thyssen Hanschel regarding conducting a field trial with fuming red nitric acid on an operational FOX vehicle and said they responded with some concern that it might result in damage to the GC/MS. Could fuming red notric acid (conc. >90%) damage a mass spectrometer? Be specific.
A. 5. Red fuming nitric acid could damage a GC/MS, especially one not designed to handle highly reactive materials. Damage could occur in any of the three regions of the GC, the inlet, capillary column or outlet, depending on the materials used for construction or deposited during use. Organic substances, certain metal surfaces or other materials may vigorously react with nitric acid or even nitrogen dioxide. In the mass spectrometer section, reaction at the silicone membrane or even in the high vacuum chamber is also possible.
Q.6. Does NIST maintain a spectrum standard for nitric acid? If not, why?
A.6. We have no reference spectrum for nitric acid in our collection, nor have we been able to find one in other collections or in a cursory literature survey. This absence is not as surprising as one might first imagine for such a common compound. Owing to the high reactivity of nitric acid, the reliable determination of its mass spectrum would probably require special instrumentation and substantial effort. Since mass spectrometry is not a practical method for the identification of nitric acid, there has been little motivation for making this determination.
Q.7. If a metallic container were used to store nitric acid, what kind of metal container would be required? If two open bullet holes were in the container for over 5 months, that is, if there were a continuous source of oxygen to the fuming red nitric acid, would it evaporate?
A.7. Large quantities of red fuming nitric acid are normally shipped and stored in aluminum or stainless steel containers. The volatility of this substance depends on the relative amounts of nitric acid and nitrogen dioxide present, with the normal boiling point varying from 25 C for a 45/55 wt%/wt% NO2/HNO3 mixture to 86 C for pure nitric acid. Once a sufficient amount of nitrogen dioxide has evaporated to raise the boiling point above ambient, further evaporation through holes in a large container would be slow.
Q.8. If a metal tray were used to transport red fuming nitric acid from this container to a mass spectrometer, would it react with the metal? What metals would it not react with?
A.8. As mentioned above, fuming red nitric acid is not reactive towards passivated aluminum or stainless steel (or other chromium-steel alloys). Passivation of these untreated surfaces occurs through a mild chemical reaction.
One issue, that of the detection of nitric acid by mass spectrometry, deserves further comment. Unless a mass spectrometer is specifically designed to detect highly reactive gases, it is not clear that a good quality spectrum nitric acid would be obtained. It is even less likely that nitric acid would pass intact through a GC and then through the silicone membrane inlet of the Bruker instrument to the mass spectrometer. Therefore, the introduction of nitric acid into a GC/MS may produce unpredictable results. Without examining the observed spectra and spectral comparison algorithms, I cannot evaluate the plausibility of the claimed false positive identifications.
Upon request, I can provide further details concerning any the above responses. I will also be glad to provide further technical assistance on these matters.
Sincerely,
Stephen E. Stein, Ph.D.
Director. NIST Mass Spectrometry Data Center
Chemical Science and Technology Laboratory
- admin
- Site Admin
- Posts: 36188
- Joined: Thu Aug 01, 2013 5:21 am
Return to Investigations of Government
Who is online
Users browsing this forum: No registered users and 7 guests


