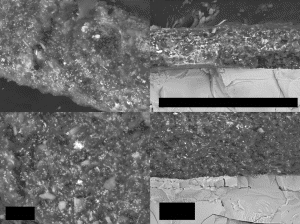
DISCUSSIONAll of the dust samples that were inspected were found to contain red/gray chips. The chips are characterized by a red layer in which XEDS analysis identifies carbon, oxygen, aluminum, silicon, and iron, and a gray layer in which mainly iron and oxygen are found. The ratios of these elements appear to be similar especially when this analysis is performed on a clean cross-section of the layers. The BSE imaging also shows the consistency of the red layers by revealing the size and morphology of the particles that are contained in the bulk of the layers. The results clearly show the similarities of the red/gray chips from the different dust samples from all four sites.
There are a number of questions raised by our results.
1. How Much of the Energetic Red Material Survived During the WTC Destruction?In the sample provided by collector J. MacKinlay the fraction of red/gray chips was roughly estimated. Fifteen small chips having a total mass of 1.74 mg were extracted from a 1.6 g sample of dust from which readily identifiable glass and concrete fragments had been removed by hand. Thus the fraction of red/gray chips was approximately 0.1% by weight in the separated dust Another sampling showed 69 small red/gray chips in a 4.9 g sample of separated dust. Further samples are being analyzed to refine this estimate. The fall of the WTC Towers produced enormous clouds of dust whose total mass is difficult to ascertain; but clearly the total mass of red/gray chips in the WTC dust must be substantial given the fraction observed in these samplings.
2. Is the Red Material Thermitic in Nature?Our observations show that the red material contains substantial amounts of aluminum, iron and oxygen, mixed together very finely. In the sample soaked in MEK, we observed a clear migration and aggregation of the aluminum away from other elements and determined that elemental aluminum and iron oxide must be present. In the product collected after DSC ignition, we found spheres which were not initially present. Many of these spheres were iron rich and elemental iron was found in the post-ignition debris. Further, the DSC traces demonstrate that the red/gray chips react vigorously at a temperature below the melting point of aluminum and below the ignition (oxidation) point of ultrafine grain (UFG) aluminum in air [18]. These observations reminded us of nano-thermite fabricated at the Lawrence Livermore National Laboratory and elsewhere; available papers describe this material as an intimate mixture of UFG aluminum and iron oxide in nano-thermite composites to form pyrotechnics or explosives [19-21]. The thermite reaction involves aluminum and a metal oxide, as in this typical reaction with iron oxide:
2Al + Fe2O3 → Al2O3 + 2Fe (molten iron), ∆H = ― 853.5 kJ/mole.
Commercially available thermite behaves as an incendiary when ignited [6], but when the ingredients are ultra-fine grain (UFG) and are intimately mixed, this “nano-thermite” reacts very rapidly, even explosively, and is sometimes referred to as “super-thermite” [20, 22].
We would like to make detailed comparisons of the red chips with known super-thermite composites, along with comparisons of the products following ignition, but there are many forms of this high-tech thermite, and this comparison must wait for a future study. Meanwhile, we compare with products of commercially available (macro-) thermite. During ignition of thermite, we have observed that many spheres and spheroids are formed as part of the molten product of the reaction is vigorously scattered. These particles tend to become spherical due to surface tension and, being small, are rapidly cooled and solidify as they fall through the air, thus their spherical shape is preserved.
To facilitate comparisons between the products of red/gray chip ignition and commercial thermite ignition, we juxtapose the respective images and XEDS spectra.
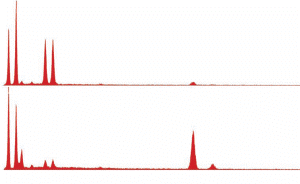
We observe that the spheroidal residues from ignition of red chips (Figs. 25, 26) possess a strikingly similar chemical signature to a typical XEDS spectrum from a spheroid generated by commercial thermite (Fig. 24). This similarity supports our hypothesis that the red chips are indeed a form of thermite.
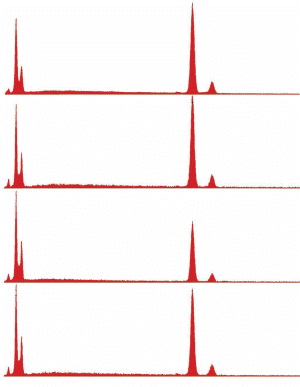
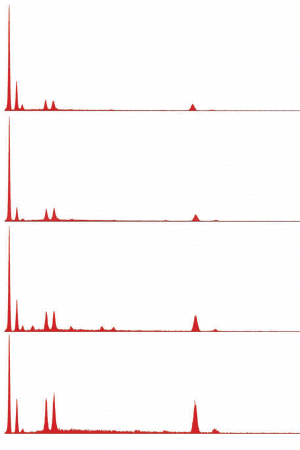
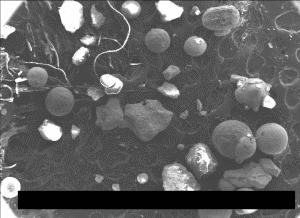 Images of spheroids
Images of spheroids 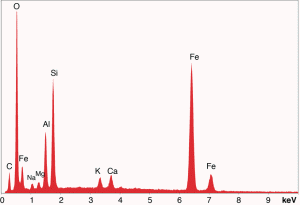
XEDS spectra of spheroids
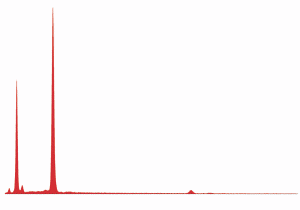
Fig. (24). Spheres formed during ignition of commercial thermite, with corresponding typical XEDS spectrum.
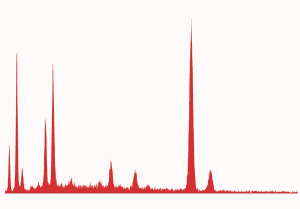 Fig. (25). Spheres formed during ignition of red/gray chip in DSC, with corresponding typical XEDS spectrum (although spheres with predominately iron and some oxygen are also seen in the post-ignition residue).
Fig. (25). Spheres formed during ignition of red/gray chip in DSC, with corresponding typical XEDS spectrum (although spheres with predominately iron and some oxygen are also seen in the post-ignition residue).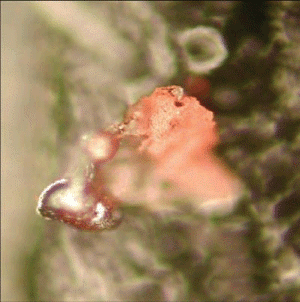
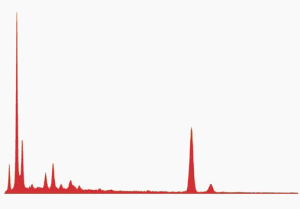
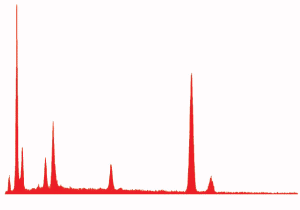 Fig. (26). Residue of red chip subjected to flame test; XEDS spectrum of left-most microsphere.
Fig. (26). Residue of red chip subjected to flame test; XEDS spectrum of left-most microsphere. 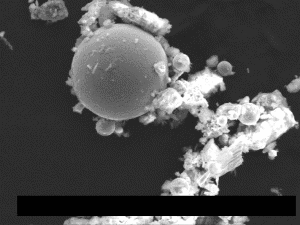 Fig. (27). Spheres extracted from WTC dust.
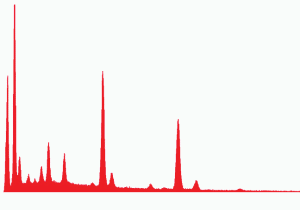
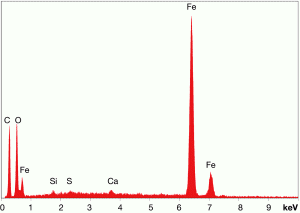
Fig. (27). Spheres extracted from WTC dust. 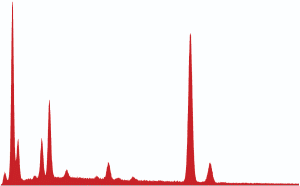 Fig. (28). XEDS spectrum from a sphere found in the WTC dust.
Fig. (28). XEDS spectrum from a sphere found in the WTC dust.In addition to the red/gray chips, many small spheres have been found by our group in the WTC dust. These contain the same elements as the residue of thermite, as noted in a previous paper [5]. We show spheres found in the WTC dust (Fig. 27) and a representative XEDS spectrum from such a sphere (Fig. 28); we invite the reader to compare these results with those found for ignition of commercial thermite and for ignition of red/gray chips (above).
3. Could the Red Material Be Unreacted “Super-Thermite”?We have noted that ordinary thermite acts as an incendiary when ignited. However, when the ingredients are ultrafine- grain and are intimately mixed, the mixture reacts very rapidly, even explosively [20]. Thus, there is a highly energetic form of thermite known as an energetic nanocomposite or “super-thermite,” composed of aluminum and iron oxide with at least one component being approximately 100 nm or less, often along with silicon and carbon [19-28].
“Reaction rates between nanosize aluminum and metal oxides can be significantly greater than those observed with traditional micron- size thermite powders. Reactions occurring between metal and metal oxide powders are accompanied by the generation of high temperatures (>3000 K). Super-thermites, formed by mixing of aluminum and metal oxide nanopowders result in energy release rate by two orders of magnitude higher than similar mixtures consisting of micron size reactants” [22].
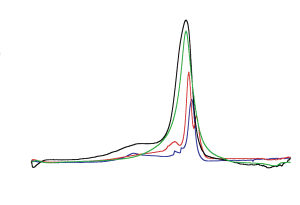
The red layer of the red/gray chips is most interesting in that it contains aluminum, iron and oxygen components which are intimately mixed at a scale of approximately 100 nanometers (nm) or less. Now we compare a DSC trace obtained for a WTC red/gray chip with a DSC trace obtained for known super-thermite (see Fig. (29)).
Ordinary thermite ignites at a much higher temperature (about 900 °C or above) and gives a significantly broader trace than super-thermite [21]. All these data suggest that the thermitic material found in the WTC dust is a form of nanothermite, not ordinary (macro-) thermite. We make no attempt to specify the particular form of nano-thermite present until more is learned about the red material and especially about the nature of the organic material it contains.
4. Did the Technology to Make Highly Exothermic Nanocomposites Exist Prior to 9/11/2001?We find the answer in a report by Gash et al. dated April 2000, seventeen months before the tragedy:
“Nanostructured composites are multicomponent materials in which at least one of the component phases has one or more dimensions (length, width, or thickness) in the nanometer size range, defined as 1 to 100 nm. Energetic nanocomposites are a class of material that have both a fuel and oxidizer component intimately mixed and where at least one of the component phases meets the size definition. A sol-gel derived pyrotechnic is an example of an energetic nanocomposite, in which metal-oxide nanoparticles react with metals or other fuels in very exothermic reactions. The fuel resides within the pores of the solid matrix while the oxidizer comprises at least a portion of the skeletal matrix.” “As an example, energetic nanocomposites of FexOy and metallic aluminum are easily synthesized. The compositions are stable, safe and can be readily ignited” [19].
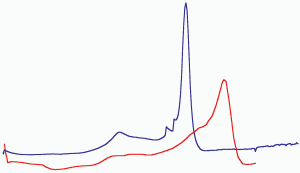 Fig. (29). DSC trace of sample 1 (blue line) compared with DSC of xerogel Fe2O3/UFG Al nanocomposite (from Tillotson et al. [28]). Both DSC traces show completion of reaction at temperatures below 560 °C.
Fig. (29). DSC trace of sample 1 (blue line) compared with DSC of xerogel Fe2O3/UFG Al nanocomposite (from Tillotson et al. [28]). Both DSC traces show completion of reaction at temperatures below 560 °C.We gather that the technology to make materials remarkably fitting the characterization of the red chips was available by April 2000. In the same report, the scientists noted that “polymers” can be added to the nanocomposite:
“This sol-gel method allows for the addition of insoluble materials (e.g., metals or polymers) to the viscous sol, just before gelation, to produce a uniformly distributed and energetic nanocomposite upon gelation. Al metal (as a fine powder, ~6μm diameter) was added to some FexOy gel syntheses just before gelation to produce FexOy /Al(s) pyrotechnic nanocomposites…. These nanocomposites were subsequently processed to make both a xerogel and aerogel of the material…. The pyrotechnic nanocomposite can be ignited using a propane torch” [19].
Indeed, the red chips can be ignited using a torch and they have properties of a pyrotechnic nanocomposite. All the required ingredients are present – aluminum, iron, oxygen, silicon, and carbon – and they are incorporated in such a way that the chip forms (and sometimes ejects) very hot material when ignited. The Gash report describes FTIR spectra which characterize this energetic material. We have performed these same tests and will report the results elsewhere. We note that polymers in the matrix may be responsible for absorption of MEK and the subsequent swelling which we observed [29].
A report on an April 2001 conference discloses who was known to be working on such explosives at that time:
The 221st National Meeting of the American Chemical Society held during April 2001 in San Diego featured a symposium on Defense Applications of Nanomaterials. One of the 4 sessions was titled nanoenergetics…. This session provided a good representation of the breadth of work ongoing in this field, which is roughly 10 years old.… At this point in time, all of the military services and some DOE and academic laboratories have active R&D programs aimed at exploiting the unique properties of nanomaterials that have potential to be used in energetic formulations for advanced explosives…. nanoenergetics hold promise as useful ingredients for the thermobaric (TBX) and TBX-like weapons, particularly due to their high degree of tailorability with regards to energy release and impulse management [20].
The feature of “impulse management” may be significant. It is possible that formulations may be chosen to have just sufficient percussive effect to achieve the desired fragmentation while minimizing the noise level.
5. Can Super-Thermite be Handled Safely?The April 2000 report by Gash et al. states:
“The nature of the wet nanocomposites also affords an additional degree of safety. In our hands, the wet pyrotechnic nanocomposites cannot be ignited until the drying process is complete. This property should allow the production of a large quantity of the pyrotechnics that can be stored safely for some time and dried shortly before its use” [19].
Safe handling of the malleable sol-gel material allows easy coating of surfaces (such as steel), which the same group, in a subsequent report, says they have achieved.
“The sol-gel process is very amenable to dip-, spin-, and spray-coating technologies to coat surfaces. We have utilized this property to dipcoat various substrates to make sol-gel Fe2O3/Al/Viton coatings. The energetic coating dries to give a nice adherent film.” “We have prepared fine powders, pressed pellets, cast monoliths, and thin films of the hybrid inorganic/ organic energetic nanocomposite” [25].
Thus, the energetic nano-composite can be sprayed or even “painted” onto surfaces, effectively forming an energetic or even explosive paint. The red chips we found in the WTC dust conform to their description of “thin films” of “hybrid inorganic/organic energetic nanocomposite”. Indeed, the descriptive terms “energetic coating” and “nice adherent film” fit very well with our observations of the red-chips which survived the WTC destruction. We cannot determine at this time, however, whether the thinness of the chips resulted from the application method or the manner of reaction. While the application of a thin film might have suited specific desired outcomes, it is also possible that the quenching effect of the steel the material was in contact with may have prevented a thin film of a larger mass from reacting. The fact that most of the chips have a distinctive gray layer suggests that the unreacted material was in close contact with something else, either its target, a container, or an adhesive.
Clapsaddle et al. further noted in their report:
“These results indicate that under ambient conditions the hybrid inorganic/organic energetic composite is very stable to impact, is spark insensitive, and only very slightly friction sensitive. As noted in the Experimental section of this report, in our hands wet hybrid nanocomposites are safe to handle and difficult to thermal [sic] ignite. However, once dry the material burns very vigorously and rapidly with the evolution of significant amounts of gaseous species” [24].
The organic component contributes to the rapid gas evolution and explosive nature of these energetic superthermites when dry [24].
“Super-thermite electric matches” have been developed at Los Alamos National Laboratory for which “applications include triggering explosives for ... demolition” [30]. It is indeed possible that such matches, which are designed to be ignited by a simple electric pulse, could contain material similar to the red material we have found in the WTC dust. With regard to the safety of super-thermite matches, the Los Alamos announcement notes:
“Unfortunately, conventional electric matches use lead containing compounds that are extremely sensitive to impact, friction, static, and heat stimuli, thereby making them dangerous to handle. In addition, these compounds produce toxic smoke. The Super-Thermite electric matches produce no toxic lead smoke and are safer to use because they resist friction, impact, heat, and static discharge through the composition, thereby minimizing accidental ignition. They can be designed to create various thermal-initiating outputs—simple sparks, hot slag, droplets, or flames—depending on the needs of different applications” [30].
6. What is the Energy Release of Super-Thermite Compared to Conventional Explosives?A graph in an article on nanostructured energetic materials [21] shows that the energy/volume yield for Al/Fe2O3 composite material exceeds that of TNT, HMX and TATB explosives commonly used in demolitions (see Fig. (30)).
It is striking that some of the red/gray chips release more energy in kJ/g than does ordinary thermite, as shown in the blue bar graphs above. The theoretical maximum for thermite is 3.9 kJ/g [27]. We suggest that the organic material in evidence in the red/gray chips is also highly energetic, most likely producing gas to provide explosive pressure. Again, conventional thermite is regarded as an incendiary whereas super-thermite, which may include organic ingredients for rapid gas generation, is considered a pyrotechnic or explosive [6, 24]. As this test was done in air it is possible that some of the enhancement of energy output may have come from air oxidation of the organic component.
7. Could the Red Chip Material be Ordinary Paint?We measured the resistivity of the red material (with very little gray adhering to one side) using a Fluke 8842A multimeter in order to compare with ordinary paints, using the formula:
Specific resistivity = RA / L
where R = resistance (ohms); A = cross-sectional area (m2); L = thickness (m).
Given the small size of the red chip, about 0.5 mm x 0.5 mm, we used two probes and obtained a rough value of approximately 10 ohm-m. This is several orders of magnitude less than paint coatings we found tabulated which are typically over 1010 ohm-m [31].
Another test, described above, involved subjection of red chips to methyl ethyl ketone solvent for tens of hours, with agitation. The red material did swell but did not dissolve, and a hard silicon-rich matrix remained after this procedure. On the other hand, paint samples in the same exposure to MEK solvent became limp and showed significant dissolution, as expected since MEK is a paint solvent.
Further, we have shown that the red material contains both elemental aluminum and iron oxide, the ingredients of thermite, in interesting configuration and intimate mixing in the surviving chips (see Results, section 1). The species are small (e.g., the iron oxide grains are roughly 100 nm across) in a matrix including silicon and carbon, suggesting a superthermite composite. Red chips when ignited produce very high temperatures even now, several years after the 9/11 tragedy, as shown by the bright flash observed and the production of molten iron-rich spheres (see photomicrographs in Fig. (20) above). Correspondingly, the DSC tests demonstrate the release of high enthalpy, actually exceeding that of pure thermite. Furthermore, the energy is released over a short period of time, shown by the narrowness of the peak in Fig. (29). The post-DSC-test residue contains microspheres in which the iron exceeds the oxygen content, implying that at least some of the iron oxide has been reduced in the reaction. If a paint were devised that incorporated these very energetic materials, it would be highly dangerous when dry and most unlikely to receive regulatory approval for building use. To merit consideration, any assertion that a prosaic substance such as paint could match the characteristics we have described would have to be accompanied by empirical demonstration using a sample of the proposed material, including SEM/XEDS and DSC analyses.
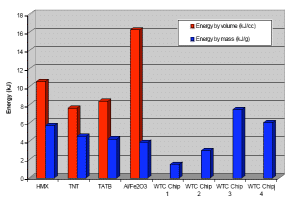 Fig. (30). Energy release for monomolecular explosives HMX, TNT and TATB, for energetic composite Al/Fe2O3, [21] and energy release by mass for four red/gray chips found in the WTC dust as measured in a Differential Scanning Calorimeter.8. What Future Studies are Contemplated?
Fig. (30). Energy release for monomolecular explosives HMX, TNT and TATB, for energetic composite Al/Fe2O3, [21] and energy release by mass for four red/gray chips found in the WTC dust as measured in a Differential Scanning Calorimeter.8. What Future Studies are Contemplated?We observe that the total energy released from some of the red chips exceeds the theoretical limit for thermite alone (3.9 kJ/g). One possibility is that the organic material in the red layer is itself energetic. Determination of the chemical compound(s) involved in the organic component of the red material would promote understanding. Further studies of the red material (separated from the gray material) compared to known super-thermite variants using DSC, TGA, FTIR (etc.) analyses would certainly be in order. In particular, NMR and GC-mass spectroscopy and related studies are urged to identify the organic material.
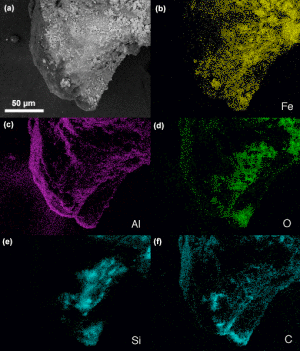
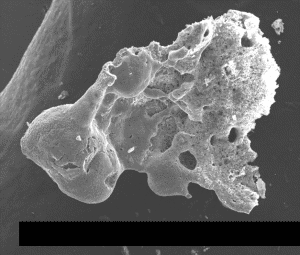
We have observed that some chips have additional elements such as potassium, lead, barium and copper. Are these significant, and why do such elements appear in some red chips and not others? An example is shown in Fig. (31) which shows significant Pb along with C, O, Fe, and Al and displays multiple red and gray layers.
In addition, the gray-layer material demands further study. What is its purpose? Sometimes the gray material appears in multiple layers, as seen in Fig. (32).
 Fig. (31). Photomicrograph of a red/gray chip found in sample 3, showing multiple layers and an unusual light-gray layer between the red layers.
Fig. (31). Photomicrograph of a red/gray chip found in sample 3, showing multiple layers and an unusual light-gray layer between the red layers.The red-mesoporous material is on the left in this view, with the touching dark-gray layer next and a lighter-gray material on the right as seen in a photograph of the same chip (right hand image in Fig. (32)). The gray layer in contact with the red layer has the XEDS spectrum shown in Fig. (33) in which iron is not seen, while the outer gray material had an XEDS spectrum just like those displayed in Fig. (6).
Thus, the middle-layer gray material contains carbon and oxygen and presumably also contains hydrogen, too light to be seen using this method. Since the gray inner layer appears between two other layers, it may be a type of adhesive, binding a red porous thermitic material to another, iron-rich material. One might speculate that the red thermitic material has been attached to rusty iron by an adhesive. The cooling effect of the iron in such close proximity, acting as a heat sink, might quench the reaction and explain the fact that unreacted red thermitic material, always found by us in thin layers, remains in the dust. These hypotheses invite further experiments.

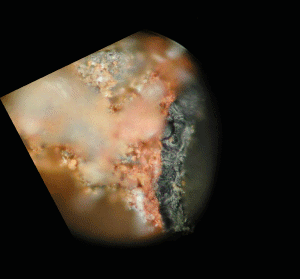 Fig. (32). Close-up SEM image of the chip pictured on the right, the same chip but not precisely the same spot. This chip had been treated in MEK solvent so that the red layer has expanded and porosity is evident.
Fig. (32). Close-up SEM image of the chip pictured on the right, the same chip but not precisely the same spot. This chip had been treated in MEK solvent so that the red layer has expanded and porosity is evident.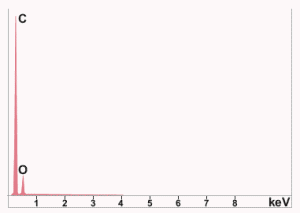 Fig. (33). XEDS spectrum for gray layer which touches the red layer of the chip shown above.
Fig. (33). XEDS spectrum for gray layer which touches the red layer of the chip shown above.No red/gray chips having the characteristics delineated here were found in dust generated by controlled demolition using conventional explosives and methods, for the Stardust Resort & Casino in Las Vegas (demolished 13 March 2007) and the Key Bank in Salt Lake City (demolished 18 August 2007). Of course, we do not assume that the destruction of the WTC skyscrapers occurred conventionally.
The red material does burn quickly as shown in the DSC, and we have observed a bright flash on ignition, but determination of the burn rate of the red material may help to classify this as a slow or fast explosive. It may be that this material is used not as a cutter-charge itself, but rather as a means to ignite high explosives, as in super-thermite matches [30]. Having observed unignited thermitic material in the WTC residue, we suggest that other energetic materials suitable for cutter charges or explosives should also be looked for in the WTC dust. NIST has admitted that they have not yet looked for such residues [11].