by Department of Health and Human Services
OFFICE OF INSPECTOR GENERAL
December, 2015
NOTICE: THIS WORK MAY BE PROTECTED BY COPYRIGHT
YOU ARE REQUIRED TO READ THE COPYRIGHT NOTICE AT THIS LINK BEFORE YOU READ THE FOLLOWING WORK, THAT IS AVAILABLE SOLELY FOR PRIVATE STUDY, SCHOLARSHIP OR RESEARCH PURSUANT TO 17 U.S.C. SECTION 107 AND 108. IN THE EVENT THAT THE LIBRARY DETERMINES THAT UNLAWFUL COPYING OF THIS WORK HAS OCCURRED, THE LIBRARY HAS THE RIGHT TO BLOCK THE I.P. ADDRESS AT WHICH THE UNLAWFUL COPYING APPEARED TO HAVE OCCURRED. THANK YOU FOR RESPECTING THE RIGHTS OF COPYRIGHT OWNERS.
Inquiries about this report may be addressed to the Office of Public Affairs at
Public.Affairs@oig.hhs.gov.
Daniel R. Levinson
Inspector General
A-06-15-00030
Office of Inspector General
http://oig.hhs.gov
The mission of the Office of Inspector General (OIG), as mandated by Public Law 95-452, as amended, is to protect the integrity of the Department of Health and Human Services (HHS) programs, as well as the health and welfare of beneficiaries served by those programs. This statutory mission is carried out through a nationwide network of audits, investigations, and inspections conducted by the following operating components:
Office of Audit Services
The Office of Audit Services (OAS) provides auditing services for HHS, either by conducting audits with its own audit resources or by overseeing audit work done by others. Audits examine the performance of HHS programs and/or its grantees and contractors in carrying out their respective responsibilities and are intended to provide independent assessments of HHS programs and operations. These assessments help reduce waste, abuse, and mismanagement and promote economy and efficiency throughout HHS.
Office of Evaluation and Inspections
The Office of Evaluation and Inspections (OEI) conducts national evaluations to provide HHS, Congress, and the public with timely, useful, and reliable information on significant issues. These evaluations focus on preventing fraud, waste, or abuse and promoting economy, efficiency, and effectiveness of departmental programs. To promote impact, OEI reports also present practical recommendations for improving program operations.
Office of Investigations
The Office of Investigations (OI) conducts criminal, civil, and administrative investigations of fraud and misconduct related to HHS programs, operations, and beneficiaries. With investigators working in all 50 States and the District of Columbia, OI utilizes its resources by actively coordinating with the Department of Justice and other Federal, State, and local law enforcement authorities. The investigative efforts of OI often lead to criminal convictions, administrative sanctions, and/or civil monetary penalties.
Office of Counsel to the Inspector General
The Office of Counsel to the Inspector General (OCIG) provides general legal services to OIG, rendering advice and opinions on HHS programs and operations and providing all legal support for OIG’s internal operations. OCIG represents OIG in all civil and administrative fraud and abuse cases involving HHS programs, including False Claims Act, program exclusion, and civil monetary penalty cases. In connection with these cases, OCIG also negotiates and monitors corporate integrity agreements. OCIG renders advisory opinions, issues compliance program guidance, publishes fraud alerts, and provides other guidance to the health care industry concerning the anti-kickback statute and other OIG enforcement authorities.
Notices
THIS REPORT IS AVAILABLE TO THE PUBLIC
at http://oig.hhs.gov
Section 8M of the Inspector General Act, 5 U.S.C. App., requires that OIG post its publicly available reports on the OIG Web site.
OFFICE OF AUDIT SERVICES FINDINGS AND OPINIONS
The designation of financial or management practices as questionable, a recommendation for the disallowance of costs incurred or claimed, and any other conclusions and recommendations in this report represent the findings and opinions of OAS. Authorized officials of the HHS operating divisions will make final determination on these matters.
EXECUTIVE SUMMARY
The Medicaid program would have received an additional $1.4 billion in Medicaid rebates on the top 200 generic drugs for 2005 through 2014 if the rebate calculation for generic drugs contained an inflationary factor similar to the rebate calculation for brand-name drugs.
WHY WE DID THIS REVIEW
The Office of Inspector General (OIG) received a congressional request to examine recent increases in the prices charged for generic drugs and the effect these prices have had on generic drug spending in the Medicare and Medicaid programs. In response to that request, OIG has updated a previous review of generic drug price increases under the Medicaid drug rebate program.
The objective of this review was to determine the extent to which generic drug price increases have exceeded the statutory inflation factor used in the rebate calculation for brand-name drugs.
BACKGROUND
The Medicaid drug rebate program became effective in 1991 (the Social Security Act (the Act) § 1927). For a covered outpatient drug to be eligible for Federal Medicaid funding, the drug’s manufacturer must enter into a rebate agreement that is administered by the Centers for Medicare & Medicaid Services (CMS) and pay quarterly rebates to the States. CMS, the States, and drug manufacturers each have specific functions under the program. Among other things, section 1927(b)(3) of the Act requires a participating manufacturer to report quarterly to CMS the average manufacturer price (AMP) and, if applicable, the best price for each covered outpatient drug.
The Act requires the payment of additional rebates for single-source and innovator multiple-source drugs (collectively, “brand-name drugs”) under certain situations. Section 1927(c)(2) requires manufacturers to pay an additional rebate when the AMP for a brand-name drug increases by more than a specified inflation factor. Generally, the amount of the additional rebate is based on the amount that the drug’s reported AMP exceeds its inflation-adjusted baseline AMP, and manufacturers pay the additional rebate for each unit of the drug reimbursed by Medicaid. The Act does not include a similar inflation-based rebate provision for generic drugs.
A previous OIG report found that generic drug price increases exceeded the specified statutory inflation factor applicable to brand-name drugs for 35 percent of the quarterly AMPs reviewed. If the provision for brand-name drugs had been extended to generic drugs, the Medicaid program would have received additional rebates. We calculated that Medicaid would have received a total of $966 million in additional rebates for the top 200 generic drugs, ranked by Medicaid reimbursement, from 1991 through 2004. We recommended that CMS consider seeking legislative authority to extend the additional rebate provisions to generic drugs. CMS agreed to consider our recommendation when it considered future legislative proposals.
HOW WE CONDUCTED THIS REVIEW
We limited our review to the top 200 generic drugs, as ranked by Medicaid reimbursement, for each year from 2005 through 2014. A total of 869 drugs were in the top 200 generic drugs at least once during the 10 years. We assigned each drug a baseline AMP based on the second quarter that pricing data was reported on CMS’s Medicaid Drug Rebate System for the drug and compared each quarterly AMP to the inflation-adjusted baseline AMP. We then applied the method in the Act for calculating additional rebates on brand-name drugs to the top 200 generic drugs for each quarter that the quarterly AMP exceeded the inflation-adjusted baseline AMP.
WHAT WE FOUND
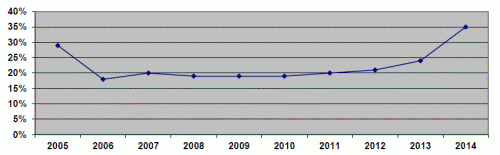
Generic drug price increases exceeded the specified statutory inflation factor applicable to brand-name drugs for 22 percent of the quarterly AMPs we reviewed. If the provision for brand-name drugs were extended to generic drugs, the Medicaid program would receive additional rebates. We calculated that Medicaid would have received a total of $1.4 billion in additional rebates for the top 200 generic drugs, ranked by Medicaid reimbursement, from 2005 through 2014. The additional rebates for the top 200 generic drugs increased most years, from more than $39 million in 2005 to more than $464 million in 2014.
CONCLUSION
Our findings are consistent with our previous work and support our prior recommendation that CMS consider seeking legislative authority to extend the additional rebate provisions to generic drugs. On November 2, 2015, the Bipartisan Budget Act of 2015 (P.L. No. 114-74) was enacted and included provisions extending the additional rebate to generic drugs. The additional rebate for generic drugs will apply to rebate periods beginning with the first quarter of 2017. Therefore, we are not making any additional recommendations.
TABLE OF CONTENTS
• INTRODUCTION
o Why We Did This Review
o Objective
o Background
Medicaid Drug Rebate Program
Previous Office of Inspector General Report on Generic Drug Prices
o How We Conducted This Review
• FINDINGS
o Generic Drug Price Increases Exceeded Inflation
o Additional Rebates
• CONCLUSION
• APPENDIX
o AUDIT SCOPE AND METHODOLOGY